Should We Be Adding HA to PRP? A Literature Review

Credit: Shutterstock
As CMO of Regenexx, it’s my job to stay up to date on the research behind the latest trends in Orthobiologics research. One of those new things is adding common knee gel shots (HA) to PRP to improve knee arthritis results more than PRP alone. So let’s explore the high-level research supporting this idea and whether it’s time to approve this practice for the Regenexx Corporate network. Let’s dig in.
PRP and HA
PRP is Platelet-Rich Plasma, an Orthobiologic that’s made from concentrating the healing platelets from your blood and in this case injecting that solution into a knee with arthritis. HA is Hyaluronic Acid, which is the stuff in a knee gel shot used to treat arthritis. It’s basically an artificial joint lubricant.
We have dozens of randomized controlled trials showing that PRP injections help patients with mild to moderate knee arthritis (click on the PDF above where each circle links to a study). HA is approved by almost all insurers as a treatment for knee arthritis based on its published data and FDA approval as a prescription drug.
The idea that PRP+HA may be helpful stems from scaffolding research that goes back almost 2 decades. Basically, this combo may create a nice matrix for new cells to grow. Hence, adding PRP to HA could be a 1+1=3 situation where the result is better than either product alone.
Why Should I Care?
I wear two hats at Regenexx. One is that I’m a practicing physician. So when patients ask me if it’s worth it to add in HA to their PRP shot, I need to know how to answer that question. The other hat I wear is that I oversee our Corporate program. That’s where over 500 employers (as of this writing) have entrusted us to add Orthobiologics to their health plan to reduce their orthopedic surgery costs and improve the lives of their employees. When I wear that hat, I’m looking at this data from the standpoint of keeping that promise to our Corporate partners and their employees.
The Research Review
I performed a search of the US National Library of Medicine PubMed system using the terms “platelet rich plasma hyaluronic acid knee osteoarthritis”. I reviewed all 239 results and found these five randomized controlled trials on knee arthritis patients:
- A 2016 RCT out of Brazil looked at 105 patients comparing PRP+HA, PRP, and HA groups and found that the combination group had the best outcomes and also reported earlier relief (1).
- A 2018 RCT from China compared 360 patients in four different treatment groups: PRP, HA, PRP+HA, and placebo. They measured a number of different metrics including serum inflammatory and cartilage breakdown markers and concluded that the PRP+HA group had the best results (2).
- A 2019 RCT out of Italy compared PRP+HA to HA alone given as three serial injections in 60 patients and found that the combination was better in most metrics at 6 and 12 months, but in some metrics, there was no difference between the groups (3).
- A 2020 RCT out of China used 122 knees (in fewer patients) and compared PRP+HA to PRP and HA alone with a 2-year follow-up. They also measured inflammatory cytokines in the knee and found that PRP+HA was superior to PRP or HA alone (4).
- A recent 2021 RCT with 84 total patients compared PRP alone to PRP+HA and found better pain relief at 6 months for the combination product (5).
My Analysis
We have 5 RCTs that I found and all conclude that PRP+HA beats either PRP or HA alone. One study was larger at 90 patients in 4 groups and the rest had a group size of between 30-60 patients each. Several studies measured inflammatory cytokines with these finding superiority of PRP+HA.
How big are these clinical effect sizes? Meaning how much of a real difference does PRP+HA make compared to PRP alone? This is a graph I created from the largest study (n=360 Yu et al):
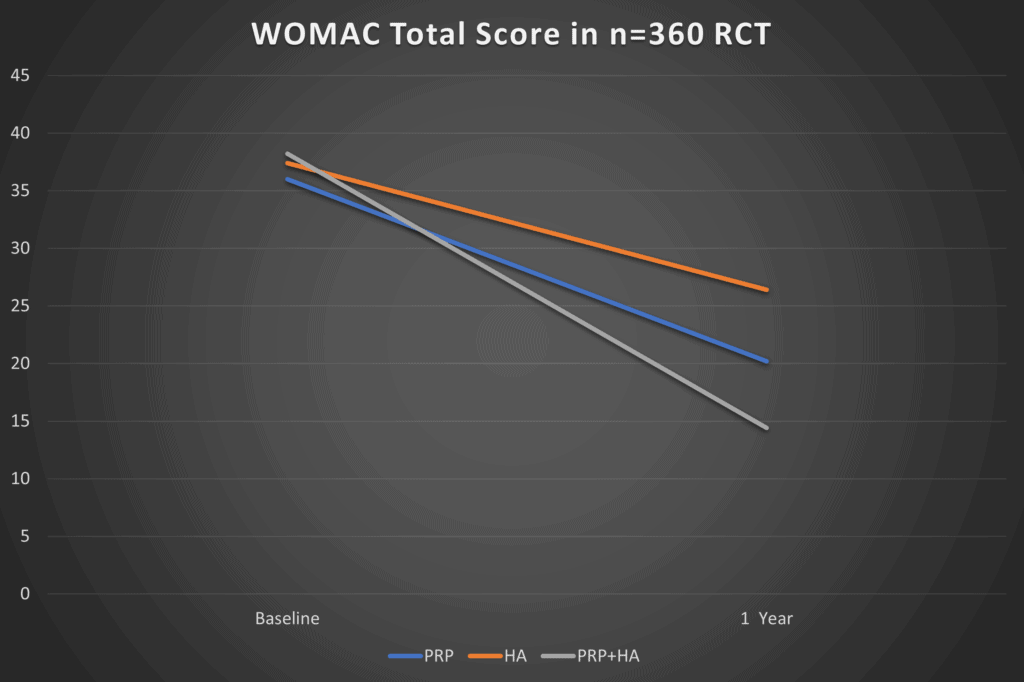
The difference between the PRP alone and PRP+HA group here is about 5 points. While that is statistically different, would the average person filling out this functional questionnaire (WOMAC) notice that 5 point difference? To get to that place, the change in this score would need to exceed the MCID for the questionnaire (Minimum Clinically Important Difference) which is 10 points (6). Hence while the additional 5 point WOMAC improvement for PRP+HA is statistically different from PRP alone, it’s not something most patients would notice.
How about the studies that show lower inflammatory cytokine levels for PRP+HA? That’s one that could be very important, after all, we know that chronic inflammation is the enemy in knee osteoarthritis by causing more cartilage degeneration and pain. That’s also a metric that’s harder to quantify, so I’ll give that edge here to PRP+HA and concede that these changes could mean that PRP+HA modifies the long-term progression of arthritis better than PRP alone. However, we have no direct clinical evidence that this happens, just lab data suggesting it might.
Conclusions?
PRP+HA looks promising. From the standpoint of asking patients whether they would like to spend the additional money to add HA to their PRP knee injections, that’s easy. You present the information and the patient decides whether it’s worth it to them or not. After all, it doesn’t increase complications.
The bigger question for me as the CMO of Regenexx is whether our Corporate program should approve the payment of additional employer money for adding HA to all knee arthritis PRP injections. To get there, we would need a Quality of Life Cost Assessment (QALY) comparing the two. Meaning, when we take this out say 5 years, does adding in the HA mean less arthritis progression over time? Does that mean fewer PRP injections for that patient over that time? Given that we don’t have this information, I can’t approve PRP+HA for use in our Corporate program at this time.
The upshot? PRP+HA looks promising and the RCT data continues to mount. The next step is a study that looks at all of this data for longer periods to determine if adding HA to PRP improves arthritis progression or causes the patient to need fewer PRP shots over time.
____________________________________________
References:
(1) Lana JF, Weglein A, Sampson SE, Vicente EF, Huber SC, Souza CV, Ambach MA, Vincent H, Urban-Paffaro A, Onodera CM, Annichino-Bizzacchi JM, Santana MH, Belangero WD. Randomized controlled trial comparing hyaluronic acid, platelet-rich plasma and the combination of both in the treatment of mild and moderate osteoarthritis of the knee. J Stem Cells Regen Med. 2016 Nov 29;12(2):69-78. doi: 10.46582/jsrm.1202011. PMID: 28096631; PMCID: PMC5227106.
(2) Yu W, Xu P, Huang G, Liu L. Clinical therapy of hyaluronic acid combined with platelet-rich plasma for the treatment of knee osteoarthritis. Exp Ther Med. 2018 Sep;16(3):2119-2125. doi: 10.3892/etm.2018.6412. Epub 2018 Jul 6. PMID: 30186448; PMCID: PMC6122407.
(3) Papalia R, Zampogna B, Russo F, Torre G, De Salvatore S, Nobile C, Tirindelli MC, Grasso A, Vadalà G, Denaro V. The combined use of platelet rich plasma and hyaluronic acid: prospective results for the treatment of knee osteoarthritis. J Biol Regul Homeost Agents. 2019 Mar-Apr;33(2 Suppl. 1):21-28. XIX Congresso Nazionale S.I.C.O.O.P. Societa’ Italiana Chirurghi Ortopedici Dell’ospedalita’ Privata Accreditata. PMID: 31168999.
(4) Xu Z, He Z, Shu L, Li X, Ma M, Ye C. Intra-Articular Platelet-Rich Plasma Combined With Hyaluronic Acid Injection for Knee Osteoarthritis Is Superior to Platelet-Rich Plasma or Hyaluronic Acid Alone in Inhibiting Inflammation and Improving Pain and Function. Arthroscopy. 2021 Mar;37(3):903-915. doi: 10.1016/j.arthro.2020.10.013. Epub 2020 Oct 20. PMID: 33091549.
(5) Sun SF, Lin GC, Hsu CW, Lin HS, Liou IS, Wu SY. Comparing efficacy of intraarticular single crosslinked Hyaluronan (HYAJOINT Plus) and platelet-rich plasma (PRP) versus PRP alone for treating knee osteoarthritis. Sci Rep. 2021 Jan 8;11(1):140. doi: 10.1038/s41598-020-80333-x. PMID: 33420185; PMCID: PMC7794411.
(6) Clement ND, Bardgett M, Weir D, Holland J, Gerrand C, Deehan DJ. What is the Minimum Clinically Important Difference for the WOMAC Index After TKA? Clin Orthop Relat Res. 2018 Oct;476(10):2005-2014. doi: 10.1097/CORR.0000000000000444. Erratum in: Clin Orthop Relat Res. 2020 Apr;478(4):922. PMID: 30179956; PMCID: PMC6259858.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.

