Alternatives to Knee Replacement Surgery for Osteoarthritis
Are You a Regenexx Candidate?Knee osteoarthritis (OA) is one of the most common orthopedic conditions. If you are experiencing OA, you understand the pain and discomfort involved and how it can negatively impact your quality of life and enjoyment of daily activities.
Historically, many patients over 40 with knee pain due to osteoarthritis (commonly referred to as arthritis) have been advised to try physical therapy, nonsteroidal anti-inflammatory medications (NSAIDs), hyaluronate or corticosteroid knee injections to reduce pain and inflammation. If these are not helpful, the next recommendation is often arthroscopic knee surgery to repair or remove any damaged cartilage or tissue that may be causing the pain. If the surgery doesn’t deliver the desired results, then total knee joint replacement is usually recommended.
Alternative to knee replacement surgery for osteoarthritis
Can Knee Surgery Reduce Pain?
Numerous studies2 evaluating the results of common orthopedic knee surgeries have shown that these procedures generally don’t work unless the patient is younger than 40 years of age. Even then, successful outcomes are not guaranteed. If this is new information to you, you are not alone. A lot of people don’t know that surgery isn’t always the solution. Read on to learn more about our regenerative medicine alternatives to knee replacement surgery for osteoarthritis.
How Does The Regenexx Approach Work For Knee Osteoarthritis?
At Regenexx, we invented a new approach to orthopedic care we call Interventional Orthopedics. This minimally invasive alternative to knee surgery uses ultrasound-guided technology to precisely inject your own bone marrow concentrate — which contains stem cells — directly where it’s needed in the joint.
The cells in your bone marrow concentrate work at the site of your injury to promote your body’s natural healing abilities and avoid surgery.
The Regenexx Approach For Knee Arthritis
During appointments, physicians in the licensed Regenexx network examine your body in motion and may use Ultrasound to observe the inner workings of the joint in real time. This gives them a much more accurate picture of what’s contributing to your pain, how function is affected, and ultimately, the root cause of the problem.
Once you’ve been evaluated, your physician will customize a treatment plan based on your specific needs. Your treatment include may include:
Regenexx SD Injectate: A patented protocol using bone marrow concentrate that contains stem cells
Regenexx SCP Injectate: A proprietary formulation of platelet-rich plasma (PRP) that’s more concentrated than what a basic bedside centrifuge machine can produce
Regenexx PL Injectate: Platelet lysate, which is a highly specialized derivative of platelet-rich plasma (PRP)
See how the Regenexx approach helped Stephanie with her chronic pain from knee osteoarthritis.
Am I a candidate?Note: Like all medical procedures, procedures using Regenexx lab processes have a success and failure rate. Patient reviews and testimonials on this site should not be interpreted as a statement on the effectiveness of treatments for anyone else.
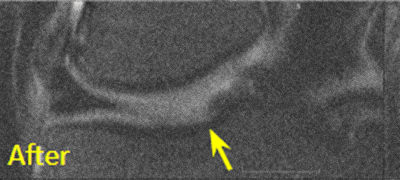
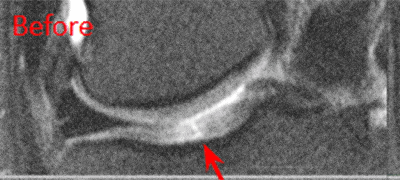
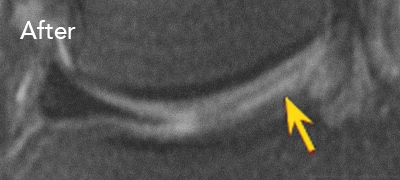
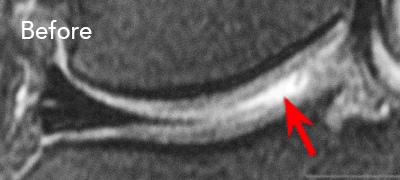
BEFORE and AFTER MRI Images
Below are the outcomes of two patients with Regenexx protocol done for knee osteoarthritis. Scroll the arrow to the right to see the MRI of the knee joint before their treatment — the white/ lighter area indicates damage. Scroll to the left to see the MRI of the knee joint after their treatment.
Patient 1 MRI: 51 years old
Patient 2 MRI: 46 years old
Patient 1: The patient was unable to return to many activities after unsuccessful microfracture surgery. As a result, underwent percutaneous, autologous, mesenchymal stem cell implant, after which they were able to return to their daily activities.
Patient 2: The patient had unsuccessful arthroscopic debridement surgery, where a large 3 cm. by 4 cm. osteochondral defect on the medial femur was discovered. The patient was then treated with percutaneously implanted autologous mesenchymal stem cells 1.5 years after surgery. The patient was able to return to full functional activities.
Am I a candidate?Patient FAQs
The knee is actually a joint — the largest one in the body. It is where the thigh (femur) and shin bones (tibia and fibula) meet. The ends of our bones are covered in a smooth, slippery material known as cartilage that helps lubricate the knee joint. This allows the bones to move against each other without friction and protects your joint from stress. Your knee also has two rings of fibrous cartilage known as the meniscus. This structure helps provide stability to the joint and distribute weight evenly.
The knee joint experiences a significant load during weight-bearing activities over a person’s life span. All joints go through normal cycles of damage and repair that can involve thinning and damaging of cartilage, bone spurs, and more, but sometimes the body’s repair process results in changes to the shape or structure of the joints. When this happens, the bones of the joint may no longer be properly aligned or lubricated, which, over time, can cause inflammation, pain, swelling, and stiffness known as osteoarthritis. Osteoarthritis can be a result of being overweight, normal aging, or an injury to the joint.
Arthritis is a general term that describes joint inflammation. There are two types of arthritis
- Osteoarthritis (OA) – also called degenerative joint disease, is the most common type of arthritis and happens when the cartilage in your joints breaks down, often in your hips, knees, and spine.6
- Rheumatoid arthritis (RA) – is an autoimmune disorder where the immune system identifies the lining of the joints (synovium) as a threat and attacks it.7
Yes. When we have looked at our data, patients who had less severe arthritis did no better than patients who had bone-on-bone arthritis. Other data published by other authors also confirm this.8
Both X-rays and magnetic resonance imaging (MRI) can show degenerative arthritis changes in the joints. However, symptoms of osteoarthritis may arise before the damage can be seen in standard X-rays. So physicians will often use the more sensitive magnetic resonance imaging, which is superior for detecting early osteoarthritis. Also, MRIs show three-dimensional images, while X-rays do not.
While cartilage is important for maximal functioning, its loss is not what causes pain. The most consistent MRI finding associated with pain is swelling in the bone called bone marrow edema (BME) or a bone marrow lesion (BML). Current research on knee pain due to arthritis has shifted from cartilage loss to this bone marrow swelling.
No, they won’t regrow large stretches of cartilage; however, they will help:
- Replace a worn-out stem cell reserve. The number of those cells available for repair and maintenance goes down as we age and get arthritis.
- Reboot dying stem cells. Studies have shown that mesenchymal stem cells can give their good batteries (mitochondria) to cells that have worn-out batteries, rebooting those dying cells.
- Treat bone lesions. Microfractures, not cartilage loss, are the source of pain.
- Reset the environment. The environment inside an arthritic joint has a mix of inflammatory and damaging chemicals.
Get started to see if you are a Regenexx candidate
To talk one-on-one with one of our team members about how the Regenexx approach may be able to help your orthopedic pain or injury, please complete the form below and we will be in touch with you within the next business day.
References
1, 4. Regenexx Patient Registry – Knee Outcomes. Lower Extremity Functional Scale (LEFS) questionnaire. Accessed on November 02, 2020.
2. Moseley JB, O’Malley K, Petersen NJ, Menke TJ, Brody BA, Kuykendall DH, Hollingsworth JC, Ashton CM, Wray NP. A controlled trial of arthroscopic surgery for osteoarthritis of the knee. N Engl J Med. 2002 Jul 11;347(2):81-8. doi: 10.1056/NEJMoa013259. PMID: 12110735. [Google Scholar]
Englund M, Guermazi A, Gale D, Hunter DJ, Aliabadi P, Clancy M, Felson DT. Incidental meniscal findings on knee MRI in middle-aged and elderly persons. N Engl J Med. 2008 Sep 11;359(11):1108-15. doi: 10.1056/NEJMoa0800777. PMID: 18784100. [Google Scholar]
Katz JN, Brophy RH, Chaisson CE, de Chaves L, Cole BJ, Dahm DL, Donnell-Fink LA, Guermazi A, Haas AK, Jones MH, Levy BA, Mandl LA, Martin SD, Marx RG, Miniaci A, Matava MJ, Palmisano J, Reinke EK, Richardson BE, Rome BN, Safran-Norton CE, Skoniecki DJ, Solomon DH, Smith MV, Spindler KP, Stuart MJ, Wright J, Wright RW, Losina E. Surgery versus physical therapy for a meniscal tear and osteoarthritis. N Engl J Med. 2013 May 2;368(18):1675-84. doi: 10.1056/NEJMoa1301408. Epub 2013 Mar 18. Erratum in: N Engl J Med. 2013 Aug 15;369(7):683. PMID: 23506518 [Google Scholar]
Sihvonen R, Englund M, Turkiewicz A, Järvinen TL; Finnish Degenerative Meniscal Lesion Study Group. Mechanical Symptoms and Arthroscopic Partial Meniscectomy in Patients With Degenerative Meniscus Tear: A Secondary Analysis of a Randomized Trial. Ann Intern Med. 2016 Apr 5;164(7):449-55. doi: 10.7326/M15-0899. Epub 2016 Feb 9.PMID: 26856620. [Google Scholar]
van de Graaf VA, Noorduyn JCA, Willigenburg NW, Butter IK, de Gast A, Mol BW, Saris DBF, Twisk JWR, Poolman RW; ESCAPE Research Group. Effect of Early Surgery vs Physical Therapy on Knee Function Among Patients With Nonobstructive Meniscal Tears: The ESCAPE Randomized Clinical Trial. JAMA. 2018 Oct 2;320(13):1328-1337. doi: 10.1001/jama.2018.13308. Erratum in: JAMA. 2018 Dec 4;320(21):2272-2273. Erratum in: JAMA. 2020 Mar 10;323(10):1001. PMID: 30285177. [Google Scholar]
3. Regenexx Patient Registry – Knee Outcomes. Numeric Pain Scale (NPS) questionnaire. Accessed on November 02, 2020.
5. Regenexx Patient Registry – Knee Outcomes. Single Assessment Numeric Evaluation (SANE) questionnaire. Accessed on November 02, 2020.
6. NIH: National Institute of Arthritis and Musculoskeletal and Skin Diseases. Accessed October 5, 2020.
7. Arthritis Foundation. Accessed November 02, 2020.
8. Xia Z, Ma P, Wu N, Su X, Chen J, Jiang C, Liu S, Chen W, Ma B, Yang X, Ma Y, Weng X, Qiu G, Huang S, Wu Z. Altered function in cartilage derived mesenchymal stem cell leads to OA-related cartilage erosion. Am J Transl Res. 2016 Feb 15;8(2):433-46. PMID: 27158337. [Google Scholar]