How does Regenexx-SD or -AD Compare to Regenexx-C?
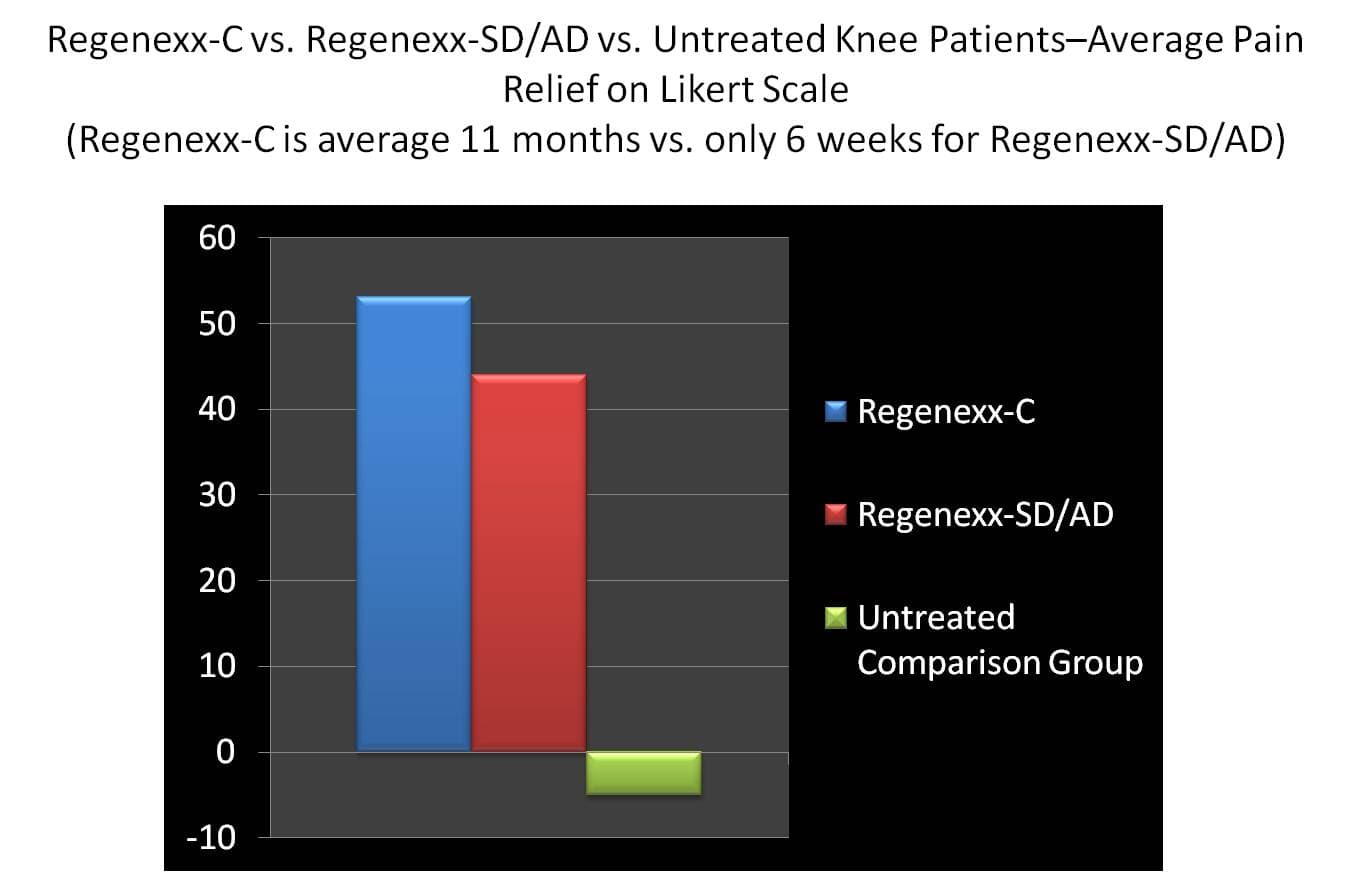
Because we have a level 3 lab as part of an interventional orthopedics practice, we offer the broadest spectrum of orthopedic stem cell and platelet based injections of any clinic. Since our Regenexx-SD and -AD stem cell injection therapies are performed in one day, they’re often more convenient for patients and are about 1/3 the cost of a cultured stem cell procedure. On the other hand, we have data on our cultured procedure going back years, cells can be saved in cryo-storage, and it produces the most orthopedic specific stem cells of any stem cell injection based procedure. As a result, we often get asked by patients to compare the results of the same day stem cell injection procedure to the cultured procedure. While we don’t have enough data on the same day procedures for a true apples to apple comparison to cultured, looking at what patients are reporting about the same day procedure is helpful. Today’s data analysis includes 140 Regenexx-SD/Regenexx-AD knee stem cell injection patients compared to 153 Regenexx-C knee stem cell injection patients. First, the graphs above and below are apples to oranges, in that the Regenexx-C patients tracked for this graph are 1-2 years out from their injection (average 11 months) and the Regenexx-SD/-AD patients are merely 4-6 weeks out from their injection. The two groups are similar in that both have a large percentage of patients who are knee replacement candidates. This high percentage of more severe arthritis patients separates this data out from published studies on PRP injections for knee arthritis, where only mild arthritis patients were treated. As you can see from above, in both procedure types (cultured vs. same day) more than half of the patients get more than 50% better. Regenexx-C has more patients who report more than 75% relief. Average relief (graph below) is similar between the two and better than an untreated group of patients who decided not to undergo either procedure (retrospectively acquired from our records and polled about a year after their decision not to have the procedure). The upshot? So far the same day procedures are hanging in there with cultured, perhaps not as stellar in the “home run” patient (>75% relief) department. What we don’t know is if the same day procedure patients will continue to improve over time, as Regenexx-C patients seem to do. On the one hand, we should have enough same day patients at 3 month and 6 month time points by late summer/early fall of 2011 to compare apples to apples with Regenexx-C [update 2/3/12-We have been working for the past several months to increase the amount of information available on -SD patients so that it is similar to the number of patients who reported -C outcomes. While taking longer than expected, we should have this comparison out soon.]
As always, it’s important to note that we know of no other clinic that looks at it’s data the way we do and reports that back to it’s patients. This is all -SD/-AD knee data, the “rock star” patients that exceed our expectations averaged in with the patients that didn’t respond. It also includes patients that had multiple procedures who initially reported a lesser outcome, but who later reported a better outcome. So what you’re looking at is quite different than a doctor stating that stem cells=magic pixie dust. Rather than magic, these adult stem cells are merely body parts to be used by physicians as tools. We believe they can be used to prevent the need for more invasive surgery. Not everyone responds, but as an investigational therapy that can help patients stay away from invasive knee procedures like knee replacements and other big surgeries such as micro fracture or ACI/MACI, we continue to be impressed.

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.