SI Joint Fusion Results and Review
This week I’ve been exchanging emails with a surgeon who does SI joint fusions. It’s been interesting as I’ve been seeing more and more advertising by a company pushing expensive, minimally invasive sacroiliac fusion devices. The surgeon didn’t like a post I did while back on the SI joint fusion technology, so I decided to take another look at SI joint fusion results. This will also be a review of SI joint fusion in general.
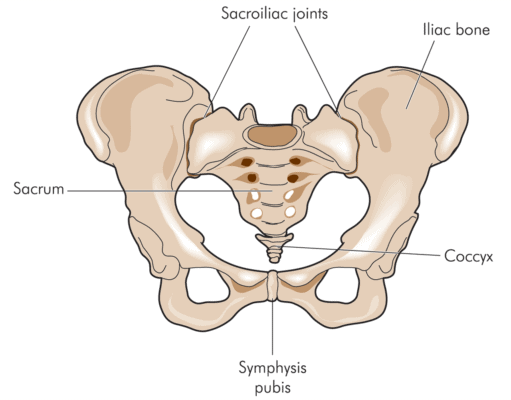
The SI or Sacroiliac Joint
First, it’s pretty funny that I’m having this back and forth with an orthopedic spine surgeon on SI joint fusion results. When I first began medical practice in the early 90s, orthopedic spine surgeons were adamant that the SI joint didn’t move. I remember many heated conversations with them that the joint also didn’t cause pain. So it’s interesting to see that a joint that didn’t move and couldn’t cause pain is now being fused because it moves and hurts too much.
Blamb/Shutterstock
The SI joint lives between the sacrum (tailbone) and ilium (back part of the pelvis) – hence the name sacroiliac. It does move, but not like a traditional joint. It gives in this direction or that and acts as an important shock absorber between the legs and the spine. Basically, it helps transmit energy from the feet to the spine and vice versa.
The joint is ear shaped and is surrounded by tough ligaments that dictate movement. The deep abdominal muscles also help to stabilize it. The SI joint can be injured by a fall on the butt, a car crash, or through other mechanisms. Pain from the SI joint generally localizes on the side of the tailbone toward the top of that structure, near the dimples of Venus (PSIS). However, it can also travel down the leg, into the groin, or to the side of the hip. Physical therapy and chiropractic are common treatments and when those fail, the next most common recommendation is to inject high dose steroids into the joint to treat chronic swelling.
Fusing the SI Joint
My SI joint fusion results review comes with a disclosure. I’ve treated patients with post fusion pain and problems my entire career. Believe me, it’s often personally upsetting to see the walking wounded these procedures produce. So I often tell my patients that fusion is a dog with big fleas. What do I mean?
Fusing any joint means that you bolt it together because it’s moving too much or is unstable. Joints that are unstable can be painful and get arthritis quicker, so at face value, it would seem that fusing them solid would be a good idea, but that’s not often the case. Don’t get me wrong, these patients typically report they’re better for few months to a few years, but then the fleas begin to bite!
The fleas are called ASD or Adjacent Segment Disease. This means that the joints above and below the fused joint begin to get painful or break down. This is caused by the fact that energy that should be handled by the fused joint gets shunted above and below to joints that are not prepared to handle those forces. So we solve one problem only to cause two new ones! This is American medicine at it’s best.
For the SI joint, the joint below is the hip and the joint above is the L5-S1 spinal segment (or the lowest part of the low back, just above the tailbone). As I said above, the SI joint is a key energy transfer mechanism, so if you fuse it, you lose that important shock absorber. What happens if you remove the shock absorbers from your car? Eventually the wheel mechanisms will be damaged. Your body is no different. The hip joint and lower lumbar spine will likely wear out faster after you remove the SI joint shock absorber.
The Research Supporting Minimally Invasive SI Fusion
My last SI joint fusion results review was about a year ago when a 6 month study seemed to show that the surgery was effective. My comment at that time was that given that most patients get bitten by the ASD fleas months or years later, a 6 month study didn’t say much. However, this was a randomized controlled trial, albeit one that was performed by a surgeon employed by the company that makes the device used to fuse the SI joint.
The new studies (see study 1 and study 2) are one and two year follow-ups of these same patients. It looks like the randomized controlled trial was abandoned at 6 months as the two new studies only report results for the treated group at 12 months and 24 months (meaning there is no longer a comparison to the patients who got physical therapy). So these new studies are fine, but not high level studies. I will review the two year report for ease of discussion.
The two year study again seems to report good results for patients who had on average about 5 years of SI joint pain before the procedure and who were diagnosed via an image guided numbing injection. However, how good is debatable. For example, before the procedure 76% of the patients needed to take narcotics for SI pain and two years after the procedure, 55% were still taking narcotics. That’s still more than half of the patients! The study reports about 5% of the patients had serious side effects, but is that number for real? Another new study, not sponsored by the manufacturer, refutes that data.
The new study is the only one I’m reviewing here today that wasn’t not paid for by the company. In fact, it’s a search of an insurance company database of the claims and complications paid by Humana on more than four hundred SI joint fusion procedures. At 6 months, the complication rate in this study was more than three times higher than that reported in the device company sponsored study (4.7% vs. 16.4%)! In addition, the company sponsored study never mentioned the fleas, nor did it have a way to measure them. The insurance study reports that a significant number of patients are reporting new spinal problems (the fleas) after the surgery. Again, fusion is a dog with fleas!
Why This is Likely an Unnecessary Surgery
When all you have is a hammer, everything looks like a nail. Meaning that surgeons tend to try to find surgical solutions for problems rather than non-surgical. Chronic SI joint pain is no different. Let me explain.
Prolotherapy (prolo) is a simple regenerative medicine technique that’s been used since the 1940s to treat SI joint pain. Because it has no business model (i.e. a hyper-expensive drug or screw that can garner huge reimbursement), there has never been the big money needed to get this simple treatment approved by insurance companies. As an example, the makers of the SI joint fusion device look like they have already spent many millions on studies, payment to surgeon consultants, and lobbying to get insurers to begin to approve and pay for this device.
Prolotherapy treatment for the SI joint would involve injecting the loose ligaments that caused the problem in the first place with the goal being to tighten those, rather than fusing the joint together so it no longer moves. The interesting thing is that there already is a randomized controlled trial comparing prolotherapy to a steroid injection of the SI joint showing similar results to the studies paid for by the SI joint device company. While it involved fewer patients, it followed patients for much longer in a controlled trial (15 months vs. 6 months) than the SI joint fusion device. Even though this was not the best form of prolotherapy (the authors tried to keep the techniques similar between the steroid and prolo groups so they just injected the prolo solution into the joint rather than into the ligaments), it still showed stark superiority over the steroid injection.
When I began performing prolo injections in these patients more than 25 years ago, I also saw nice results. However, when more advanced regenerative medicine (regen med) options became available like platelet rich plasma and same day stem cell treatments, I moved many patients to those treatments and have never looked back. In addition, as our clinic’s techniques evolved to recognize the different areas that needed to be treated in many of these patients that were outside of the SI joint, our results improved. Suffice it to say that I haven’t seen an SI joint patient in years that I felt needed this surgery. In fact, I only know of one of our patients out of more than 100 primary SI joint pain patients in the last few years who opted for this procedure despite regen med treatment. Hence, while there may be a handful of patients who need the SI joint fusion procedure, they are few and far between. My biggest concern is that the company making this device is aggressively positioning it as a good solution for every patient who doesn’t get better with physical therapy and who responds to a numbing injection. That would be a real tragedy, as I can’t imagine how many new walking wounded we’ll see over the next decade who seemingly did well for a year or two after an SI fusion, only to need a hip replacement or low back fusion because of overloaded joints elsewhere.
The upshot? The SI joint fusion results from the new research are no more compelling than the old research. While there may be patients out there who can’t be treated in any other way, pushing an SI joint fusion for most patients with chronic SI joint pain is not a good idea. However, with the many millions this company is spending on getting insurers to pay for this surgery, I expect we will see a whole new generation of walking wounded. Just remember, once you go down this route, this procedure can’t be undone, your natural SI joint is lost forever. In addition, please research other options such as prolotherapy or newer regen med options such as PRP or stem cells from a provider who routinely and successfully treats SI joint pain and knows the procedural tricks to get the best results.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
