Dr. Tami and the Seattle Stem Cell Center Review
What happens when a functional medicine and aesthetic practice begins using minimally invasive procedures to treat a panoply of incurable diseases? That’s what I’ll explore today by using the Seattle Stem Cell Center as an example, but frankly, this issue is so common these days that it could literally be any of hundreds of clinics. Let’s dig in.

The Stem Cell and Exosome Wild West
Most of us would take for granted that there’s a minimal level of physician expertise for common medical procedures. Meaning, doctors go through extensive rigorous training that results in board certification and while some physicians may be more expert than others, for the common stuff there’s a minimum competency level. Hence, it doesn’t make much of a difference who does your procedure. While that would be true for most things in medicine, there’s a stark contrast when we get to stem cell and exosome procedures. This review will help you understand what I mean.
Dr. Tami and the Seattle Stem Cell Center

Right up front on Dr. Tami’s Seattle Stem Cell Center website, we see a video that states that she is a “Double Board Certified MD”. So that’s where we’ll start our review. While I was able to confirm that she is a licensed physician, I was unable to confirm much else.
For example, I did find a very skeletal CV online which lists many certifications that are basically courses for specific skills in aesthetic medicine, acupuncture, etc.., but there is no board certification listed. I then went to ABMS, the organization that keeps records on physician board certification and I was unable to find ANY active board certification.
Dr. Tami’s CV claims that she is board certified in:
- Aesthetic Medicine (not AMA recognized)
- Integrative and Holistic Medicine (not AMA recognized)
Note that she places the disclaimer “Not AMA recognized”. That’s because neither of these listed board certifications is anything like an actual board certification that your family, pain, or orthopedic doctor has earned. So Let’s dig in here a bit.
What is an Actual Board Certification?
Getting an actual board certification is rigorous. Let’s look at what it takes to get an actual board certification (on average):
- A grueling internship year of working 60-80 hour weeks
- 3-4 years of residency training in a university hospital at 50-70 hours a week
- A written board certification exam
- An oral board certification exam
That’s just under 14,000 hours of training. If you go onto to fellowship or are in some board certification scenarios, then there are even more hours.
Now let’s look at one of Dr. Tami’s “Board Certifications” in aesthetic medicine. The American Academy of Aesthetic Medicine states that the candidate takes 56 hours of courses and then a 6 month period of gaining experience in your office and then takes a test. So here’s the difference in a graph in the training requirements for actual board certification and what Dr. Tami claims is a board certification (I gave Dr. Tami an extra 100 hours for some experience in the clinic over 6 months):
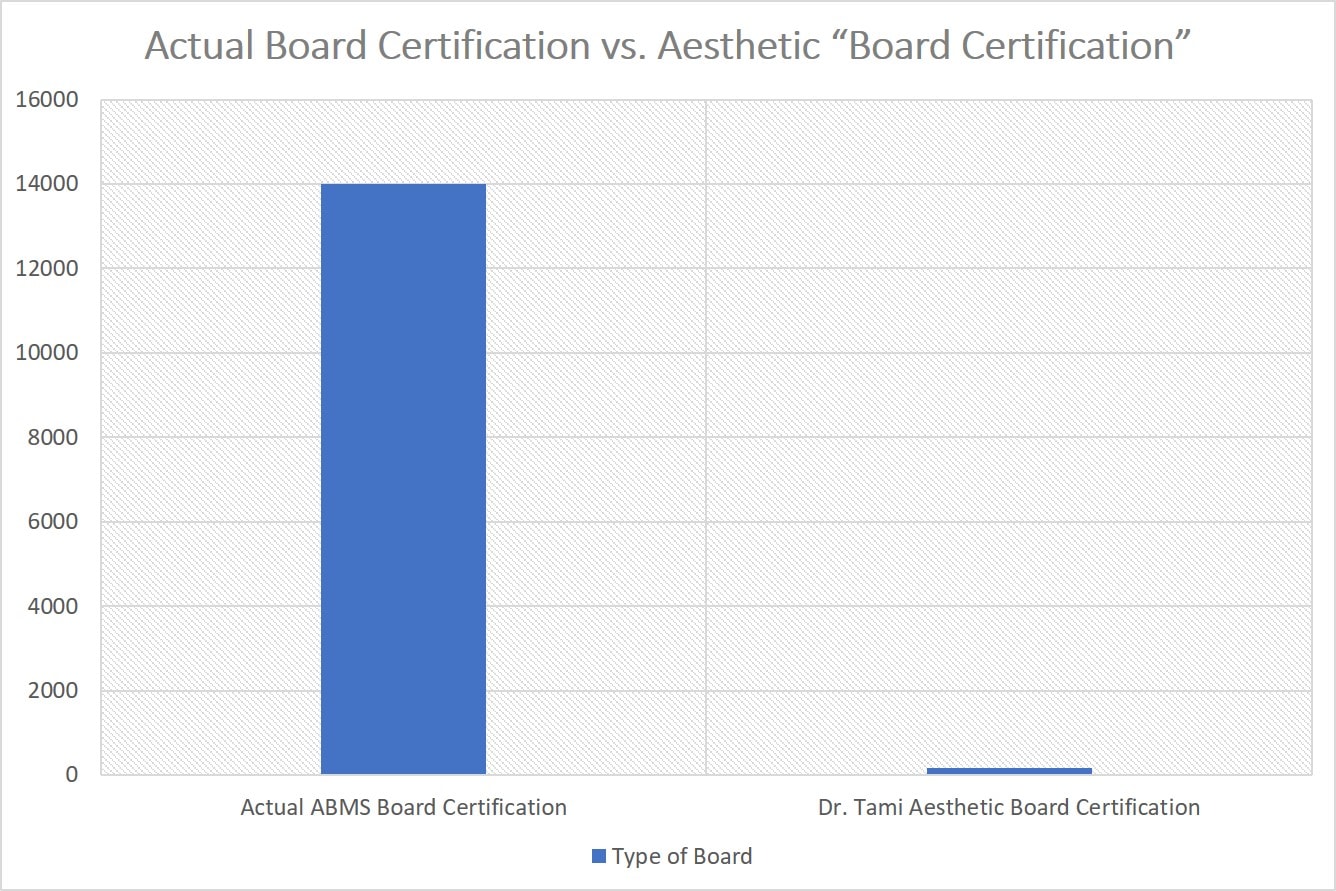
So as you can see, when Dr. Tami’s video claims that she is “Double Board Certified” that’s a pretty big stretch. She had a tiny fraction of the training required by the ABMS to call yourself “Board Certified”
Treating Everything
So now that we know that Dr. Tami never completed any board certifications like those for pain medicine, physical medicine, family practice, neurology, orthopedics, etc… let’s look at what the Seattle Stem Cell Center treats. After all, she only claims to have some expertise in Aesthetic and Functional medicine, but those aren’t actual board certifications recognized by any hospital or the American Board of Medical Specialities.
35 different conditions are listed on her website from MS to ALS to knee pain to erectile dysfunction. These medical conditions fall into the following medical specialties (board certifications):
- Orthopedics/Pain Medicine/Physical Medicine and Rehab
- Neurology
- Urology
- Cosmetics/Plastic Surgery
- Cardiology
- Pulmonology
- Gastroenterology
- Endocrino0logy
- Immunology
Dr. Tami holds none of these board certifications. So a legitimate question is what is a physician without ANY formal board certifications doing treating patients with severe incurable diseases. Or on the back, neck, knee, shoulder, etc…what is she doing treating those conditions. Let’s explore that last topic a bit more.
Spine Treatments
One of the places to separate the proverbial stem cell wheat from the chaff is usually when physicians claim to perform spinal injections but have no formal training in interventional spine. Dr. Tami’s Seattle Stem Cell Center website clearly states that she treats back and neck issues, but she has no training in image-guided injections of the spine on her CV. So what is she doing? See my animated image below:
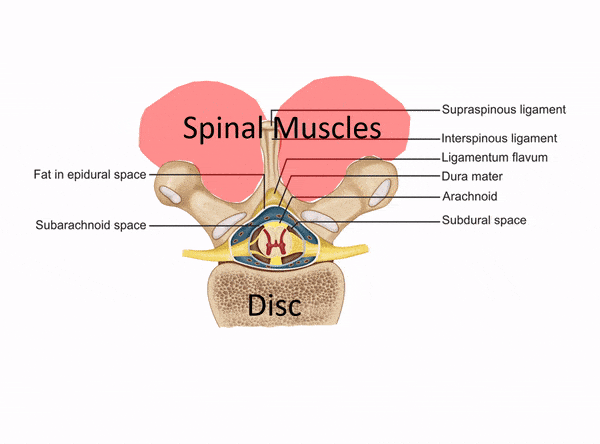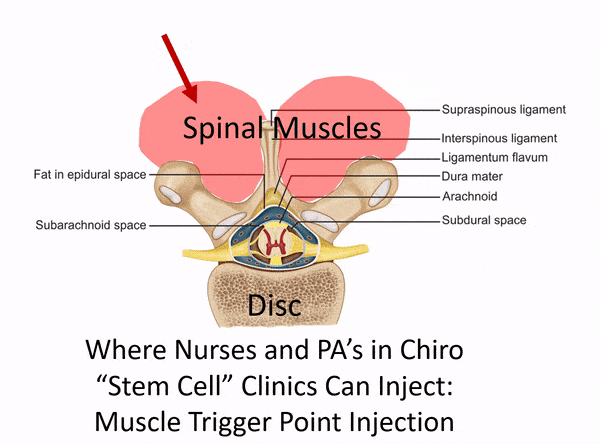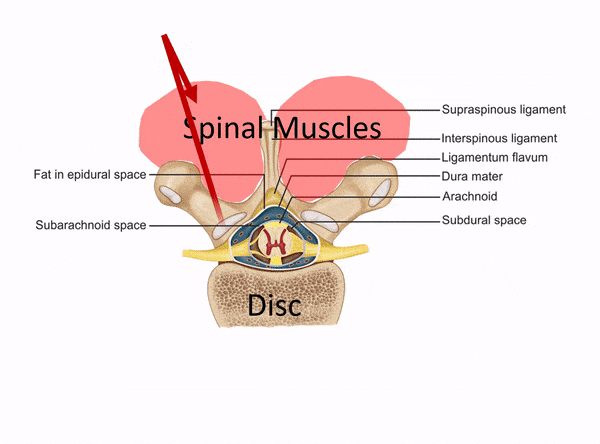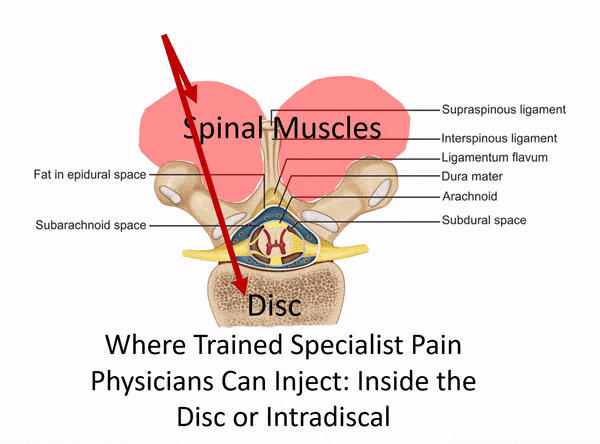
Without interventional spine training (meaning training in how to inject deep structures in the spine with x-ray guidance), all Dr. Tami can inject are the superficial muscles and ligaments (which is the same as a nurse or PA in a chiropractic clinic). What if you need stem cells in your deeper facet joints, around specific spinal nerves, or into the disc? That’s not going to happen given Dr. Tami’s training.
Cell Surgical Network
Dr. Tami is part of the Cell Surgical Network, a chain of clinics that uses adipose stromal vascular fraction (fat stem cells). The main clinic site for Cell Surgical Network was raided a few years back by the FBI for using an illegal smallpox virus. The FDA has also filed an injunction to shut the whole CSN network down and that case is pending in the California courts. Part of the issues brought up with CSN were around IRB approvals and since Dr. Tami proudly lists an IRB approval number on the Seattle Stem Cell Center web-site, let’s dig into that topic.
The IRB Approval
What is an IRB? That stands for Institutional Review Board. It’s basically a way to protect patients who are involved in clinical research. However, Cell Surgical Network got into hot water because, in order to study fat stem cells, the FDA requires a separate drug trial approval for each clinical indication. So that means that in addition to having IRB approval, you also need to have an FDA IND (approval to perform a drug study). CSN had IRB approvals, but no IND approval.
To get a sense of what CSN was missing, here that would be 35 different FDA studies. Not even Pfizer has enough money to fund that many studies. So the IRB number on Dr. Tami’s website (ICSS-2016-004) refers to a study that requires FDA approval but has no such approval. In fact, if the California courts find for the FDA (likely), then in the next few weeks to months that and the other twenty-something studies that CSN claims to be performing will be shut down by the feds.
Birth Tissue Stem Cells and Exosomes
Dr. Tami’s video on her website front page also discusses birth tissue stem cells and exosomes. Dr. Tami claims that she uses Wharton’s Jelly products that have immune-privileged stem cells. She also claims that exosomes don’t have any genetic material or DNA. So let’s dive deeper here as well.
As I have blogged extensively, studies performed by our lab, Cornell, and CSU show that the commercially available amniotic and umbilical cord products available to Dr. Tami have no living stem cells (5-7). So her claim that she’s using umbilical cord stem cells is not supported by the laboratory evidence that we have. In addition, the FDA just put out a warning on this stem cell source after companies continue to make false claims that they are selling umbilical cord stem cell products.
Then we have exosomes. Dr. Tami claims that she uses them to enhance the effectiveness of her treatments and basically describes how you can “upgrade” (my paraphrase) your procedure by adding exosomes. So do we have any clinical evidence that exosomes will, in fact, help any of the conditions that Dr. Tami treats? Nope. Not a single clinical study is published. Finally, the FDA just put out a warning on four patients in Nebraska who were made sick by exosome products and again reiterated that any such products would require FDA approval, and no such products have that approval. So if Dr. Tami is using exosomes, that’s an illegal drug product without a shred of published clinical evidence of efficacy.
Dr. Tami also claims that exosomes have no genetic material. In fact, just the opposite is true. Exosomes contain mRNA and DNA (1-4). Hence exosomes do have loads of genetic material. In fact, when they contain DNA, that’s often not a good thing.
Is Dr. Tami Using a Second Illegal HCT/P?
A video on Dr. Tami’s site was very concerning. CSN had previously been involved with American Cryostem, which was illegally culture expanding fat stem cells and then banking them. ACS received an FDA warning letter 2 years ago. Dr. Tami has a video in which she says, “…the fat has a little bit more (stem cells), but when you’re banking, that is not a problem because the bank takes care of the numbers for you.” That sure sounds like the “bank” is growing the cells in culture, which would be illegal in the U.S.
The Healing Miracle and Dr. Tami
Dr. Tami’s website touts that she’s in an episode of a Docuseries called “The Healing Miracle”. Hence, she must be a stem cell expert, no? Well, as I have shown in many blog posts, that docuseries was created by ex-con Brent Dedilitch and Jeff Hayes and was a marketing venture that was later tied to a fake Facebook patient group, a call center, and a referral service (Stem Cell Hotline). More importantly, I showed that many of the “experts” in the film weren’t actually experts in any traditional sense of the word.
If we dive into the idea of expertise in medicine, what defines it? Publications in peer-reviewed journals, presenting research data at scientific conferences, book chapters, etc… If I search the US National Library of Medicine I can find no studies on stem cells authored by Dr. Tami. There are also no medical book chapters. There are no scientific data presentations. She does have a bunch of videos, podcasts, and consumer-focused books she’s written. So in my opinion, Dr. Tami doesn’t meet the standard of a medical expert in regenerative medicine.
A Word on Functional Medicine Clinics and Stem Cells
I generally like the idea of functional medicine. I believe that traditional medicine is not always the best option. In addition, for some patients having the ability to access treatment via diet, exercise, and supplements is a great alternative to some of the crazy medications pharma shoves down our collective throats. Having said that, when these clinics add stem cells, in my opinion, they all tend to have similar problems to Dr. Tami:
- Little expertise in the use of regenerative medicine
- Providers without rigorous board certification
- Treating all sorts of incurable diseases outside of their area of expertise
- Making claims that can’t be verified
Meaning, in my opinion, very few functional medicine clinics are doing this work at a high level. Other specific problems include making claims that the umbilical cord products they’re using have young and vital stem cells (which we know is not true), and many are now using illegal exosome products without any evidence that these actually work.
The upshot? This takes a bunch of time! To dig deep into these clinics is a big job. Why do it? Because it’s really hard for the average patient to really understand the difference between high-quality orthobiologics care and low-quality care using regenerative medicine. In this case, in my opinion, with Dr. Tami, we have more the latter than the former.
_____________________________________
References:
(1) Thakur BK, Zhang H, Becker A, et al. Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res. 2014;24(6):766–769. doi: 10.1038/cr.2014.44
(2) Kalluri R, LeBleu VS. Discovery of Double-Stranded Genomic DNA in Circulating Exosomes. Cold Spring Harb Symp Quant Biol. 2016;81:275-280. doi: 10.1101/sqb.2016.81.030932.
(3) Sansone P et al. Packaging and transfer of mtDNA via exosomes. Proceedings of the National Academy of Sciences Oct 2017, 114 (43) E9066-E9075; DOI: 10.1073/pnas.1704862114
(4) Fernando MR, Jiang C, Krzyzanowski GD, Ryan WL. New evidence that a large proportion of human blood plasma cell-free DNA is localized in exosomes. PLoS One. 2017;12(8):e0183915. Published 2017 Aug 29. doi: 10.1371/journal.pone.0183915
(5) Berger D, Lyons N, Steinmetz, N. In Vitro Evaluation of Injectable, Placental Tissue-Derived Products for Interventional Orthopedics. Interventional Orthopedics Foundation Annual Meeting. Denver, 2015. https://interventionalorthopedics.org/wp-content/uploads/2017/08/AmnioProducts-Poster.pdf
(6) Becktell L, Matuska A, Hon S, Delco M, Cole B, Fortier L. Proteomic analysis and cell viability of nine amnion-derived biologics. Orthopedic Research Society Annual Meeting, New Orleans, 2018. https://app.box.com/s/vcx7uw17gupg9ki06i57lno1tbjmzwaf
(7) Panero, A, Hirahara, A., Andersen, W, Rothenberg J, Fierro, F. Are Amniotic Fluid Products Stem Cell Therapies? A Study of Amniotic Fluid Preparations for Mesenchymal Stem Cells With Bone Marrow Comparison. The American Journal of Sports Medicine, 2019 47(5), 1230–1235. https://doi.org/10.1177/0363546519829034

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
