What if the Sopranos Created an Umbilical Cord Product?
This has been an interesting month or so since the beginning of the FDA enforcement period. We’ve seen countless birth tissue companies take their amniotic and umbilical cord products off the market for fear of FDA enforcement. You would think that this would mean that all of these companies would be running for the hills. Think again. Let’s revisit Vitti Labs, who are IMHO, the Sopranos of the umbilical cord industry.

Credit: Shutterstock
Selling Umbilical Cord “Mesenchymal Stem Cells” into the Middle of an FDA Crackdown
My last name is Centeno, which many believe is Italian, but it’s actually Venezuelan. However, I have always loved Italy and the Italian people. However, with a name like Vitti labs, the first thing I thought of when I saw what this company was doing is a Tony Soprano award. Why? Because if you continue to violate the clear directives of the FDA by selling a Wharton’s Jelly product that you claim has mesenchymal stem cells in the summer of 2021, AFTER the FDA enforcement began on May 31st, you may be the reincarnation of Tony Sporano himself.
Our Research
While the publication has been delayed because of COVID, our paper looking at the stem cell content of umbilical cord products claiming that they have stem cells is almost out. Our research group found no live and functional stem cells in any product tested. For more info, see my video below:
The Email
I get sent stuff all the time and this was an email sent by a sales rep for a company called Vitti Labs to a colleague on July 11th:
“Extensive Medical is the marketing and wholesaling arm of Vitti Labs. We are proud to say that we represent over 700 active practices using Vitti Labs products and are excited to invite you to learn more about how we can help you get started.
We offer a variety of perinatal products such as Wharton’s Jelly (mesenchymal stem cells), Nano-pods containing Growth Factors, Nucleic Acids and Peptides.”
So despite the research showing that commercially available Wharton’s Jelly products have no live and functional mesenchymal stem cells and despite the FDA crackdown on umbilical cord companies selling products purporting to have mesenchymal stem cells, these guys are still at it.
Well, maybe they’re not making any efficacy claims for this product? Because that would dig this proverbial grave deeper. Then I found this:
“All these products have been used to treat thousands of patients with amazing results.”
and this…
“We have one practitioner that had been charging $2,000 to $3,000 for sexual health shots – he now charges only $700 because he can see so many more patients per hour.”
What are “sexual health shots”? Usually injections into the vagina or penis. Ouch!
Vitti Labs
Likely in a bid to avoid FDA scrutiny, Vitti seems to have pulled all claims off of its website about its “WJ-Pure+” product. It also should be noted that Vitti Labs doesn’t manufacture anything, but instead is a reseller of someone else’s products:
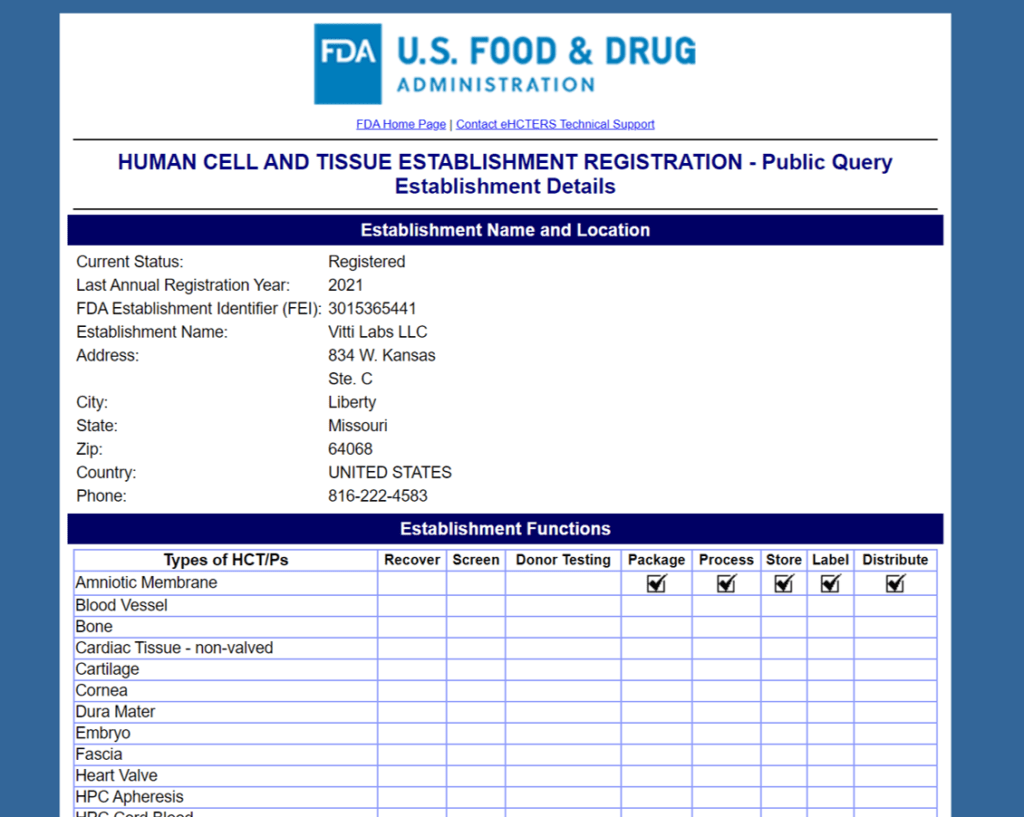
The upshot? Why is any company sending emails to providers claiming to sell umbilical cord stem cells in the summer of 2021? Your guess is as good as mine. However, I will give Vittl Labs my Tony Soprano award or in Italian, “Ne hai di fegato!”

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
