More Exosome Insanity: Genisent Labs and Extensive Medical Marketing
I get sent things all the time. This past week I was sent flyers on yet another”exosome” product made by Genisent Labs and this one came with actual treatment protocols. You just can’t make this stuff up. Let’s dig in!
What are Exosomes?
Exosomes are the latest buzz word in regenerative medicine. The concept is that stem cells excrete little packets of information to tell other cells how to behave. These include exosomes. To learn more, see my video below:
Do we have any clinical data showing that exosomes as being sold by this or any other company are effective for any clinical indication for which they’re being marketed? Nope. Hence, exosomes are all fluff right now with little clinical reality. This is why, despite producing trillions of exosomes each week at our licensed culture expansion site in Grand Cayman, we flush the exosomes down the drain and use the stem cells.
The Marketing Materials
I get sent marketing materials that violate FDA regulations every week. Hence, those produced by Extensive Medical Marketing and Genisent Labs are par for the course. However, what I was sent was also unique. Why? The marketing materials also included treatment protocols for everything from knee arthritis to orgasm improvement.
On the latter note, this was in the materials under “Male Sexual Health”:
“Grasp and pull the head of the penis toward you. While maintaining light tension, select a site for injection..with a steady and continuous motion, penetrate the skin with the needle at a 90-degree angle…advanced to the hub. Slight resistance is encountered as the needle passes through Buck’s Facia into the corpus cavernosum”
This is just one of many protocols in that document that utilize the company’s exosome product. In these marketing materials, the exosome product has clinical indications ranging from bicipital tendonitis to arthritis to erectile dysfunction (male sexual health) to Autism to Parkinson’s Disease.
Why is a Medical Marketer Distributing Treatment Protocols?
We already know that claiming to sell exosomes is not legal, as multiple medical providers including myself have pinged the FDA TRIP program and asked this question. The FDA states that exosomes require full clinical trials and drug approval, taking many years per clinical indication to ensure safety and efficacy. Instead, what companies like Genisent and Extensive Medical have done is to skip that step and instead incorrectly register the product on the FDA’s free Tissue Establishment Registration system.
However, in my opinion, the distribution of medical treatment protocols ups the ante a bit. Meaning, not only is the product an unapproved drug, but the seller has claimed numerous medical indications for that drug and provided specific treatment protocols including dose, patient prep, injection, and aftercare instructions. So while a physician at a medical conference may say here’s how I use this product or a drug manufacturer might tell doctors about the protocol they used for their FDA approval trial, a salesperson providing a medical treatment protocol to a doctor is atypical.
Eureka, an IRB Will Protect Me!
First, in my opinion, if you’re a doctor and you are silly enough to allow a salesperson with no medical training to give you a treatment protocol to treat a patient, you’re likely not providing high-quality medical care. Hence, you would likely be the type of physician who would fall for this next dodge. This is what I call the IRB two-step!
The marketing materials from Genisent and Extreme Medical also contain an IRB document from an organization calling itself Pathway Research Institute. This statement, which is in the document, is just about the worst misrepresentation of IRB and FDA regulations that I have ever seen. In fact, in my opinion, this statement is dangerous:
“Our mission revolves around the advancement of regenerative medicine research and alternative treatments or products including cellular and acellular products, ozone, peptides, and vitamin therapy to name a few. We do this by creating more opportunities for great minds and dedicated teams to validate their work through the traditional, FDA approved pathway of an IRB.”
Why is this a problem? This last sentence makes it seem like this IRB provides an alternate FDA approval pathway. Is this true?
What is an IRB?
IRB stands for Institutional Review Board. It’s basically a panel of mostly physicians, but also scientists, attorneys, ethicists, and community members who review research to protect patients. What most physicians don’t comprehend is that the FDA process and an IRB are really two separate things and that using an IRB without also having an FDA approval to conduct research confers no regulatory protection whatsoever.
There are two types of IRBs. The first is a community IRB that you might find at your local hospital. The second is one that is designed to oversee FDA approval studies. The latter has what’s called an FWA which stands for Federal Wide Assurance. The statement above suggests that Pathway has an FWA. Do they? Here’s the look-op from HHS:
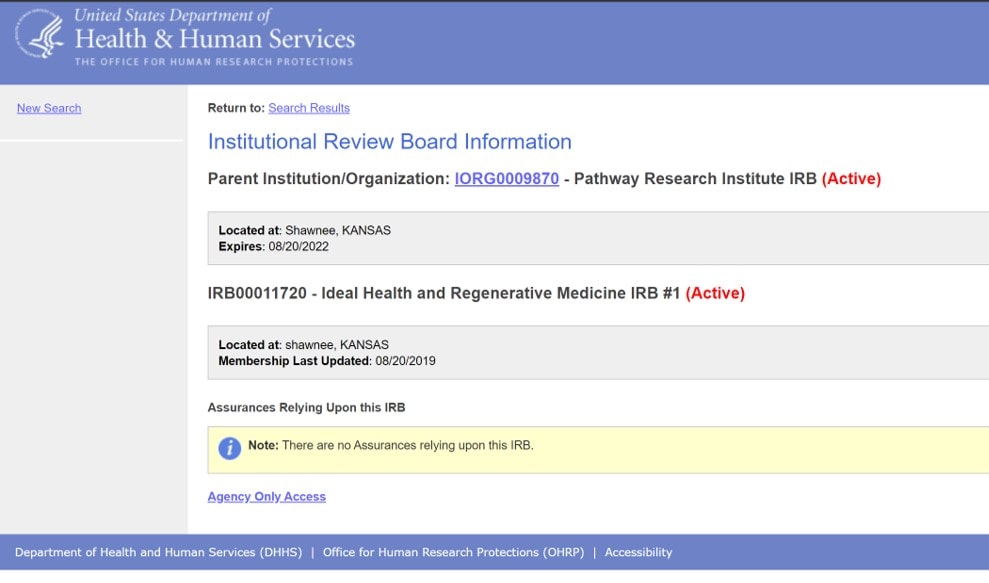
In the yellow highlighted area we see that Pathway Research Institute has no FWA. Hence, it’s a statement that it’s IRB has anything to do with FDA regulation is false. In fact, just to study an exosome product you would need much more than Pathway’s IRB, you would need an FDA IND/BLA, which is an FDA approval to begin a drug trial. Just having Pathway’s IRB in place confers no regulatory protection whatsoever. In fact, in my opinion, what the FDA would do here is not only nail you for using a drug product without approval, but they will also nail you for using an IRB without an FWA when one was required.
Genisent Labs and Pathway Research Institute-Is There a Connection?
On the top of the exosome clinical protocol sheet are websites listed for Extensive Medical and Vittilabs. Extensive Medical’s website is still under construction. Vittilabs has no website at this point. However, the chairman of the Pathway Research Institute is no other than Phillip Vitti. In addition, both Genisent Labs and Pathway are located in Shawnee, Kansas. So is there a connection?
Who is Genisent Labs?
Genisent Labs lists three employees on Linkedin. A DME sales guy, an athletic trainer, and a computer consultant. James Moberg, with degrees in Architecture and Economics and some medical sales experience, is listed as the co-founder. The company was founded this year (2019). They also sell Wharton’s Jelly which they claim is a stem cell product. We now know from multiple FDA letters to manufacturers that you also can’t make this claim. Further, through multiple independent studies, we know that this claim is also not accurate, meaning commercially available Wharton’s Jelly products contain no live and functional MSCs.
The upshot? While I’ve become used to companies selling exosomes and violating FDA regulations, this one is unique. In my opinion, not only are there FDA violations here but the addition of non-physicians distributing treatment protocols where multiple medical indications are listed is really interesting from a regulatory standpoint. In addition, misrepresentations about the protections afforded by an IRB and possible connections between the IRB reviewing the use of the products and the companies selling the products is also disturbing.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
