Mesenchymal Stem Research Continues at a Furious Pace

How important are mesenchymal stem cells (MSCs) in the big scheme of things? What are they? I’ve published a few blogs on the pace of MSC research through the years, so let’s dig in.
Mesenchymal Stem Cells
If there was one stem cell in your body that was the all-around and versatile decathlete, it would be the MSC. First, they’re in many of your tissues including your bone marrow, fat, knee fluid, knee fat pad, and elsewhere. Second, they can differentiate into many of the cells of your body such as muscle, cartilage, bone, ligament, tendon, fat, etc… That means they can replace damaged cells. Third, they can help other cells repair tissue by issuing chemical commands, acting as a general contractor in the repair response. Fourth, they can reboot dying cells by transferring their good batteries (mitochondria) to those cells.
These past few years there’s been a push to rename them “Medicinal Signalling Cells”, however, from looking into the prevalence of that term in the U.S. National Library of Medicine, it looks like this hasn’t stuck. Why the suggested name change? When the MSCs don’t come from your own body, they tend to be killed off by the killer T part of the host’s immune system, so any actions that they have are only through their ability to secrete chemical commands to other cells. Hence, in this situation, they don’t differentiate into other cells. However, when they’re from your own body, they do stick around, differentiate into other tissues, and engraft into the area.
The State of the MSC Research
I graphed the number of publications per year using the search phrase “mesenchymal stem cells” that were indexed in the US Library of Medicine (PubMed) and here’s what I got:
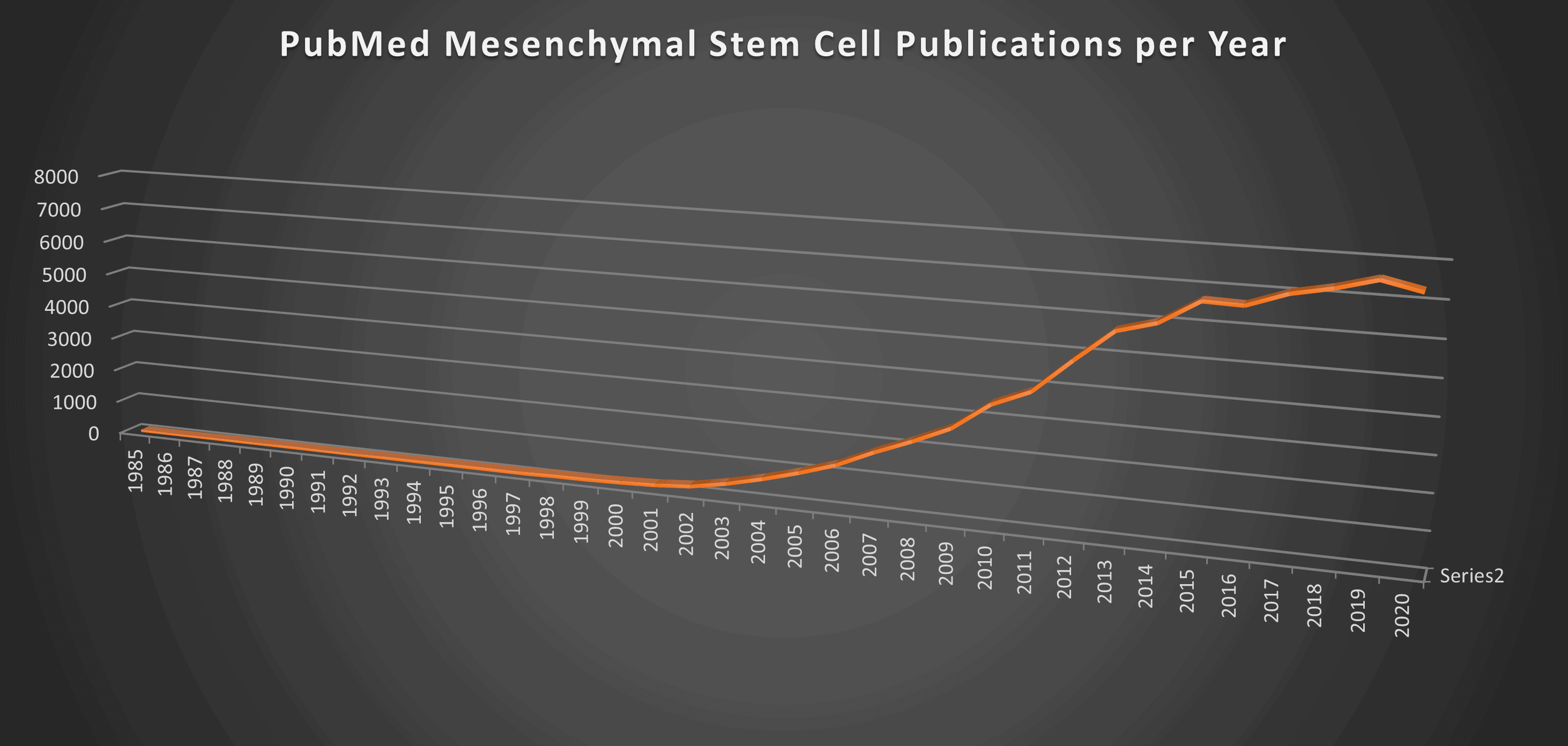
The MSC research really started to get rolling in the early 2000s which is when we got involved (2005). By the second decade of this new century, the research built quickly and in 2017, it hit the milestone of 7,000 papers a year being published. It’s still clicking along at that level.
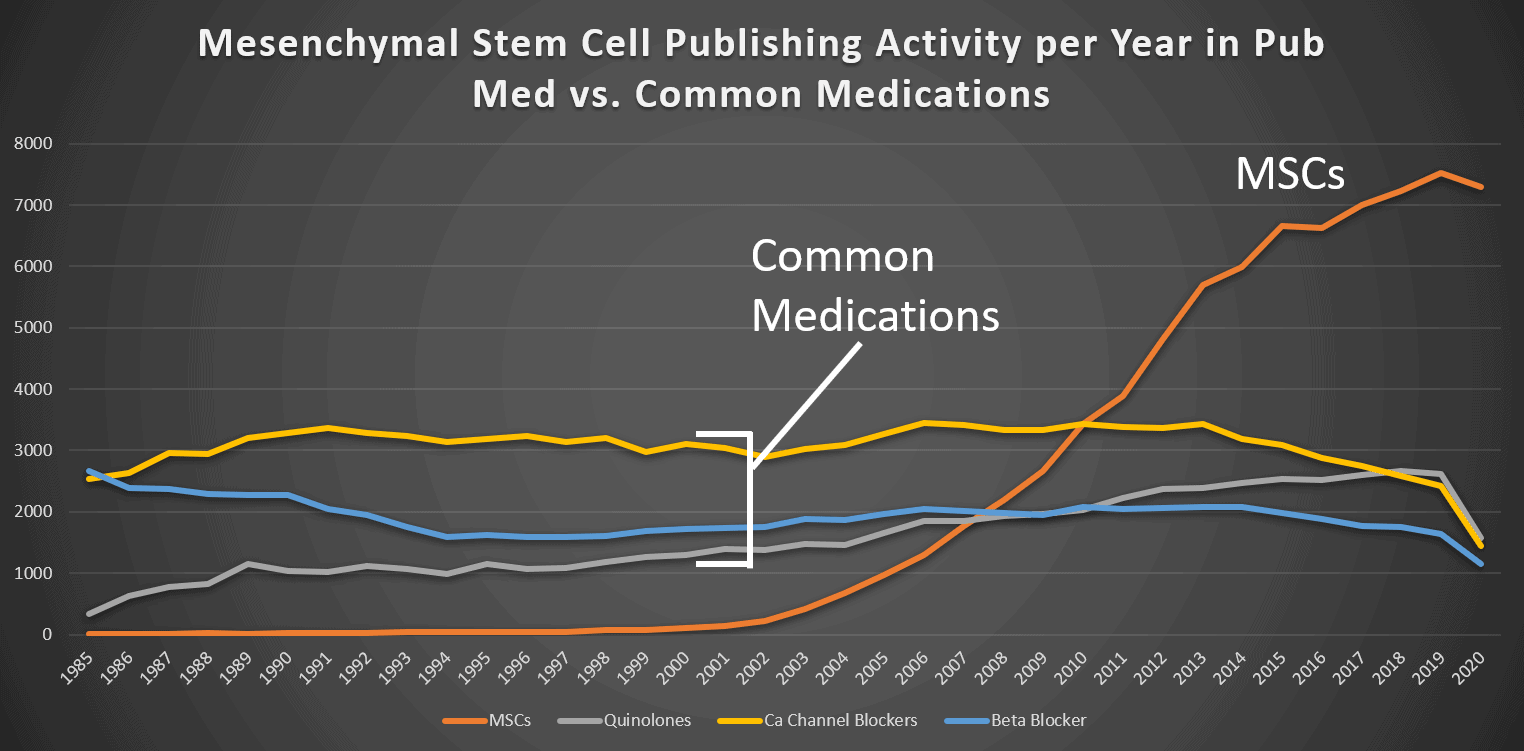
Let’s put that number in perspective by comparing this research activity to common traditional drug classes. Below I graphed publishing activity for MSCs and three common drug types. Note that in 2010, MSC research surpassed all three drug types. In 2020, there were 7,292 papers published on MSCs. Compare that to 1,582 papers in Pubmed that were indexed under the term “quinolones” that year. In addition, under 3,000 papers a year have been published for the drug types “calcium channel blocker” and “beta-blocker” (common drugs used in cardiac care) over the past decade.
How does MSC research activity compare to other common stem cell types being researched for treatment? In 2020, 2,408 papers were published on embryonic stem cells and 2,589 papers were published on induced pluripotent stem cells. Hence, MSCs are still king of the stem cell research game.
Our Research on MSCs
What has made Regenexx unique is that unlike many other companies in this space, we perform basic science research and clinical research. How many other physician network groups have this capability? None of which I’m aware. How many tissue manufacturers? A few have hired physicians to perform some clinical research or hired labs to create some white papers (many of which I have shown are misleading), but none have the ability to perform basic science or clinical research in house. How about device manufacturers like the companies that make bedside centrifuge kits? They too lack the ability to perform basic science or clinical research in-house.
On the basic science side, what has our lab research team published these past few years? Here are some examples:
- The effect of local anesthetics on mesenchymal stem cells
- How the concentration of PRP impacts tendon cells
- A method for looking at MSC dose after bone marrow concentrate treatments have been performed
- The MSC content of various commercially available umbilical cord products-submitted for publication
How about our clinical research? Those papers are too numerous to list, but here’s that link. Suffice it to say that we aim to publish 2-3 papers a year.
The upshot? MSC research is still king with more than 7,000 papers a year being published. Regenexx has always done its fair share of that research!

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
