Can the Organelles in Our Cells Get Injured? Mitochondrial Rescue
You know that your body has organs and that these can be injured by trauma. But did you know that your cells have organs that can be damaged the same way? Today I’ll review a new field of stem cell biology that may well form the basis for why some of our existing treatments work and lead the way to improved future therapies. To do that, we’ll focus on the batteries that power your cells, called the mitochondria.
Organs and Organelles
You know that your body has organs like the liver, spleen, heart, and lungs. However, did you know that your cells also have organs? Because they’re tiny, scientists have given them the name of organelles (literally little organs). Organelles include the mitochondria, endoplasmic reticulum, nucleus, Golgi body, etc… Today we’ll focus on the mitochondria.
Mitochondrial Batteries
In your cells are organelles called mitochondria which act as the energy supply or cell batteries. They turn chemicals into stored energy through the creation of a molecule called ATP. Without this energy supply, your cells quickly die.
Can Organelles be Injured by Force?
I’ve been blogging all week on the lectures from the Havemeyer orthopedic stem cell think tank conducted at CSU. One of the more interesting presentations was by Michelle Delco of Cornell. One of her areas of research is how blunt force trauma to cartilage cells impacts the mitochondria and how best to fix these issues to treat damaged cartilage.
It’s been known for quite some time that blunt force trauma to cartilage can injure the chondrocytes (cartilage cells) which then die off and leave a hole. How that happens is not completely known. Dr. Delco presented convincing evidence that force can injure the mitochondria. This is not different in principle than a person being involved in a car crash and rupturing their spleen. In this case, images were shown of damaged mitochondrial after impacting equine cartilage cells during arthroscopic surgery.
Can We Fix Dying Mitochondria?
If force can cause the batteries in your cells to get damaged, can they be fixed? While there may well prove to be chemical ways to do this, one of the more interesting phenomena that has been known about for a while is mitochondrial rescue via stem cells. How does that work?
A long time ago I did a little video on mitochondrial transfer as one of the ways in which stem cells help repair damaged tissue. Watch my video below to learn more:
Dr. Delco’s Research on Mitochondrial Transfer
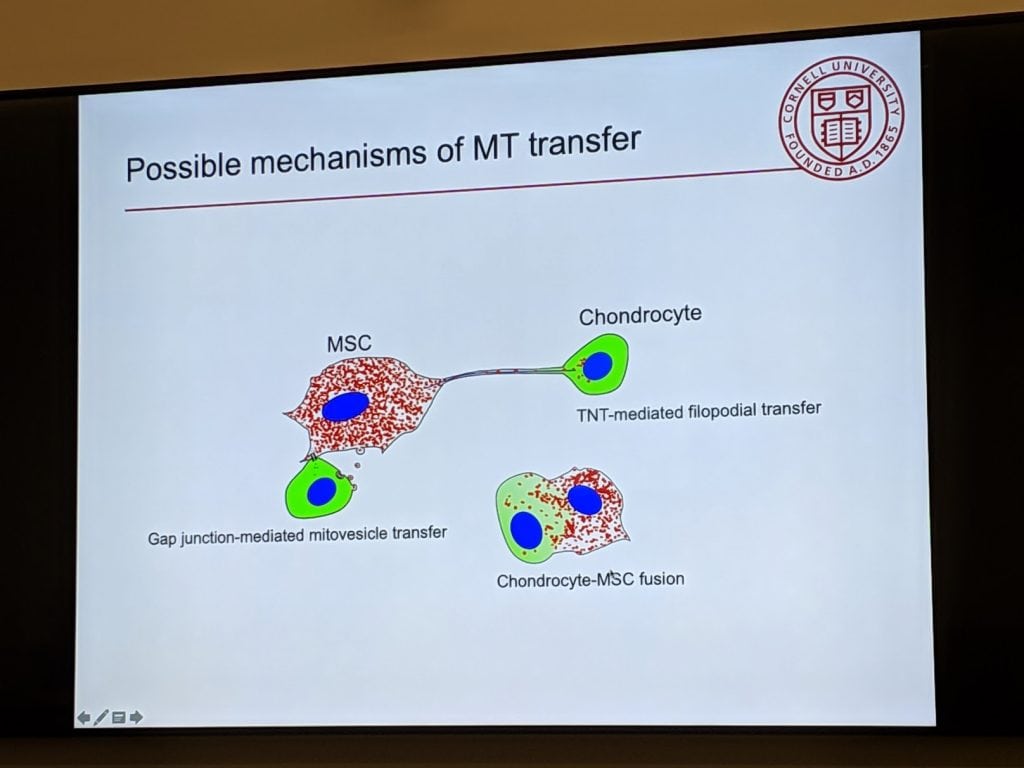
In one of the slides from her talk above, there are several ways that stem cells can transfer their good mitochondria to cartilage cells that are dying because of damaged mitochondria. They can build little tunnels (filopodia), they can package mitochondria into vesicles (exosomes), and they can fuse with the cartilage cells and transfer them that way.
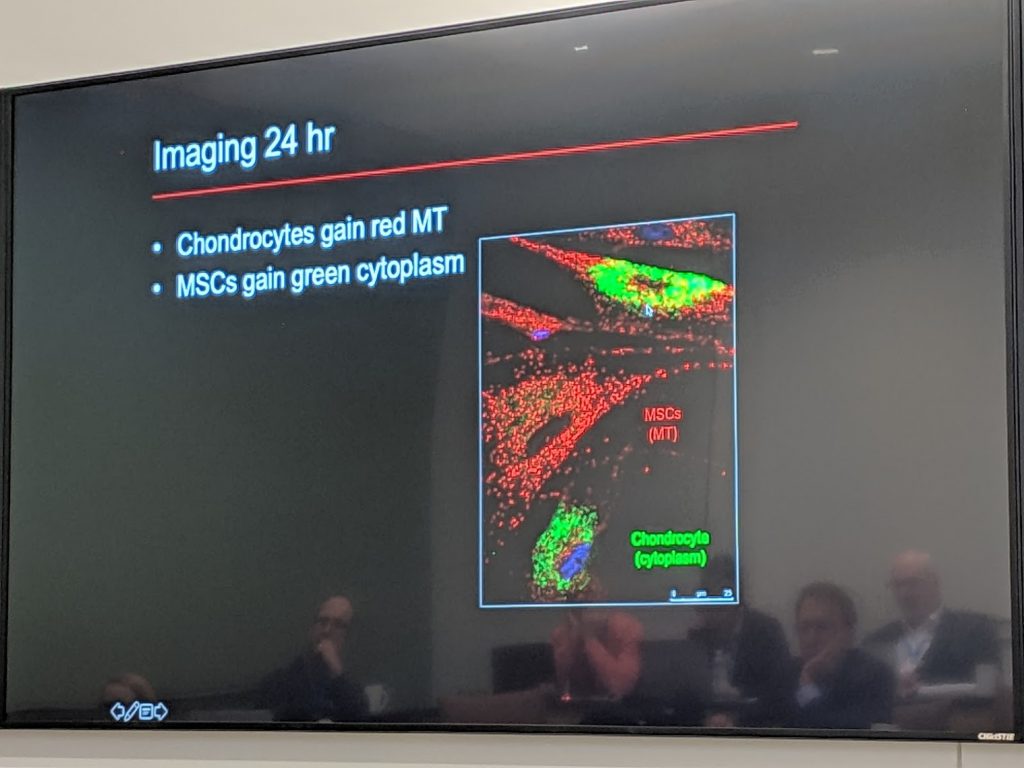
Here’s another slide using fluorescent microscopy (z-stack) that demonstrates that the stained red mitochondria from mesenchymal stem cells have been transferred into an injured cartilage cell (chondrocyte).
More Research on this Topic
There’s quite a bit published about mitochondrial transfer in mesenchymal stem cells. Take this study which demonstrated that bone marrow mesenchymal stem cells (MSCs) could transfer their mitochondria to damaged nerve cells in an animal model of spinal cord injury. Or this research that demonstrated that MSCs could rescue damaged intervertebral disc cells through many mechanisms including transferring their mitochondria. Or finally, this paper that demonstrated that MSCs could rescue damaged vascular cells in an animal model of stroke by using mitochondrial transfer.
The Future?
Can we make mitochondrial transfer better and more efficient? Are certain stem cell types better at this than others? Can we produce drugs that will heal damaged cell batteries? Can you find other ways to dump good mitochondria into damaged cells? Can gene therapy help design cartilage cells that have more robust mitochondria? Lots of research to be done.
The upshot? Mitochondrial rescue by transferring good mitochondria from stem cells to injured cartilage cells is likely happening in the real stem cell therapies in use today. In this case, there’s an argument for using stem cells very soon after cartilage injury, to save cells before they die off. Perhaps in the future, there will be other ways to prevent these damaged cell batteries from going belly up.

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
