Hand Arthritis Surgery Alternative: Which Works Better-Cultured or Same Day Stem Cells?
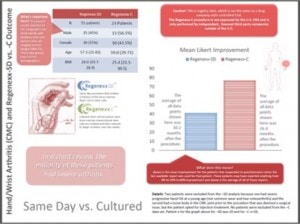
Are one type of stem cells a better hand arthritis surgery alternative than the other? I’ve been continuing our data analysis of 2013/2014 patient results using our advanced treatment registry, now moving onto comparing patient results in certain body areas by stem cell procedure type. In this infographic above (released yesterday), our results with same day stem cells (Regenexx-SD) is compared to cultured stem cells (Regenexx-C-prior Colorado and Grand Cayman experiences combined). I used the last available Likert improvement score for both mean improvement measures. While most of the differences between the two procedures are more subtle and less pronounced, this difference is stark. This is consistent with our patient experience. Why do cultured stem cells tend to work so much better in hand arthritis and show less of a difference in knee arthritis? We’re still not sure. The effects could be due to the fact that we still have a smaller numbers of patients treated for hand arthritis versus knee arthritis, so the difference could get smaller as the numbers get bigger. However, I tend to doubt this will happen, given we’ve gained clinical experience using both types of cells for many years. The upshot? If you have more severe hand arthritis, then a cultured stem cell procedure where cells are grown to bigger numbers over a number of weeks seems to be a better choice than a same day stem cell procedure. As always, click on the thumbnail above to see the bigger PDF of the treatment registry document.
The Regenexx-SD procedure is used in the U.S. (same day), while the cultured stem cell procedure (Regenexx-C) is not U.S. FDA approved and is only used via license to companies that operate outside of the U.S. for use wholly in a country that allows such use.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.