How Another Bad Surgical Orthobiologics Paper Can Teach Us About QC
Credit: Shutterstock
If there’s one journal that keeps regularly getting it wrong on orthobiologics, it’s the journal Arthroscopy. So let’s dig into a recent paper published in that journal. By understanding why it can’t support its conclusions we’ll learn quite a bit about the quality control of a bone marrow aspiration. Why? Because if you’re a doctor or patient, good or poor outcomes from a bone marrow concentrate procedure are often determined by how much time your doctor takes with this step. Let’s dig in.
Low versus High Volume BMAs
It’s hard for patients to understand that there are stark differences in the quality of medical care. Why? Because what physicians do is complex and if a doctor decides to skip a few steps in a procedure and this reduces the efficacy of that intervention, how would a patient know?
All procedures using bone marrow concentrate (that contain your stem cells) begin with what’s called a bone marrow aspiration. This means the doctor places a specialized needle known as a trocar into the bone marrow and aspirates out a sample of the liquid portion (BMA or bone marrow aspirate). The cells in that sample are then concentrated via a centrifuge to produce bone marrow concentrate (BMC), which is the mix the doctor injects.
The BMA procedure can be done in two main ways:
- Hard for the doctor-multiple low volume pulls of BMA from different sites. This takes much longer.
- Easy for the doctor-one long pull of BMA from the same spot. This is really fast.
We’ve known for many years that the multi-site/low volume BMA gets many more stem cells and that the easy way (one long pull from the same spot) gets very few (1-3).

I’m a visual learner. Hence, when I see something complex and try to explain it, I often begin drawing it out. Hence, that’s what I’ll do here.
On the left, I drew a graphical representation of total nucleated cells in the bone marrow (all cells excluding red blood cells in the sample) as blue and the stem cells as red. Hence, as you can see, there are more stem cells (red) in the low volume BMA and fewer in the high volume approach. Why? Because regrettably, the bone marrow space communicates with the peripheral blood circulation, which is very nucleated and stem cell poor compared to bone marrow. Hence, if you pull a small volume you’re getting the local stem and other cells near the tip of the needle. If you pull more volume, those cells are depleted and then you’re just pulling blood from the periphery.
Now note the illustration on the far right above. This is the main problem with today’s research paper. Let’s dig into that study.
When Surgeon Convenience Trumps Good Medicine
Recently a paper was published that seemed to show that the number of nucleated cells in a bone marrow concentrate sample wasn’t predictive of the stem cell content (5). Huh? What does all of that mean? Why should you care?
When a doctor draws a bone marrow aspirate for an orthobiologic injection, as shown above, how he draws that sample is key. The goal is to maximize the number of stem cells in that sample.
The total number of cells (all circles) in the sample is called the TNCC (Total Nucleated Cell Count). Why is this important? Because, in general, in a properly drawn BMA, there is a fixed relationship between the TNCC and the number of stem cells. Hence, if we had to find a dosing metric for stem cell content of a sample, TNCC would be pretty good, as it’s easy to measure and that data can be available to the doctor performing the orthobiologic procedure. For example, our research has shown that TNCC can predict outcomes on knee arthritis procedures (6). Meaning, if the TNCC is low, the doctor needs to make specific clinical decisions to maximize the number of stem cells in a knee. For example, treating only one knee to maximize the stem cells and not treating both knees where the meager stem cell content would be diluted across two sites.
Hence, when the conclusion of this study disconnected the TNCC count from the stem cell count, that’s important. Meaning if TNCC is related to stem cell content, doctors can have an easy way to determine the dose of BMC treatments. If not, then there is no use in knowing the total number of cells in the sample.
The Really Poor BMA
Remember when I told you that I would teach you about how to quality control a BMA? Now let’s look at the images above, focusing on the right-most diagram. In this new research paper, instead of taking a proper multisite/low volume draw, my surgical colleagues (the authors of this paper), took a huge volume of BMA from a small bone in one pull. That bone was the humerus, which in its totality, only holds about 50 ml of bone marrow aspirate. Meaning, the rocket scientists here drew on average 86 ml of bone marrow from that little bone. Why? It’s easy for the surgeon.
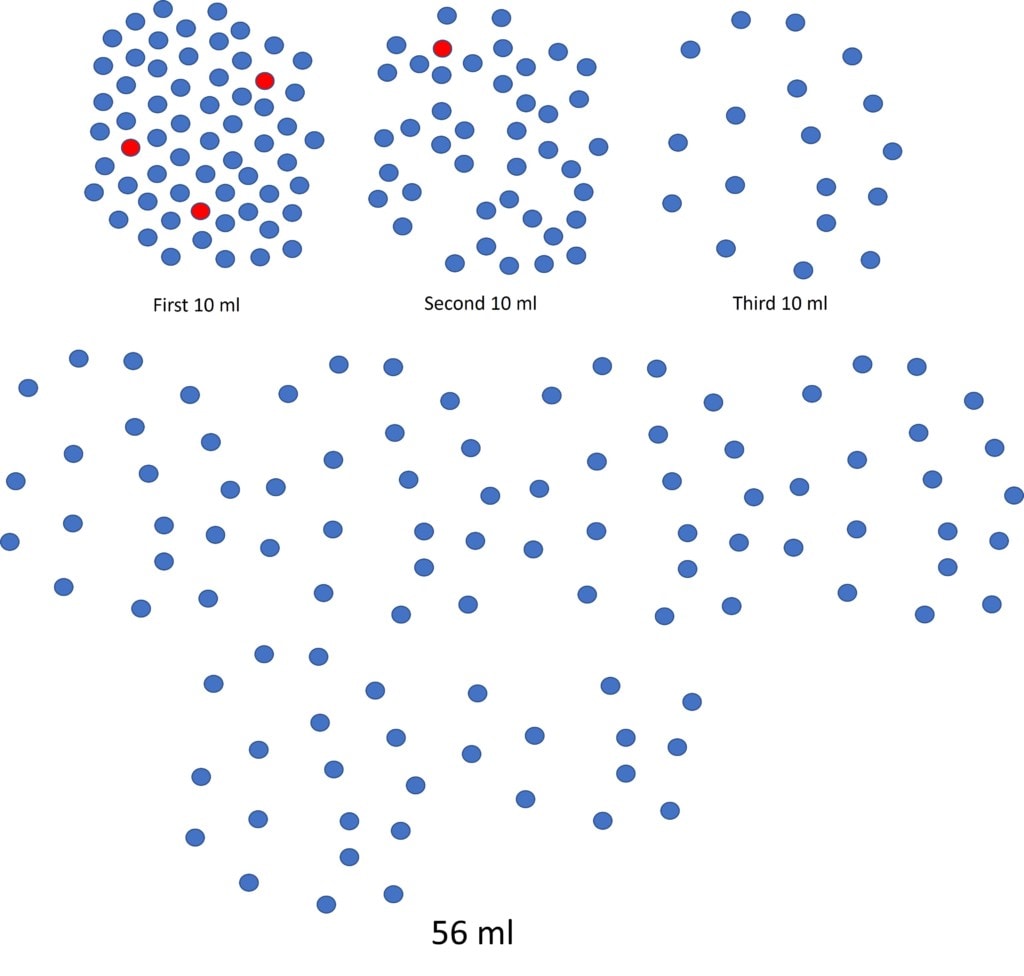
What’s more remarkable here is that the research already published by this group showed them what would happen to TNCC if they did it this way (7). The above illustration is drawn from that data in that paper (and others) (1-3). The first 10 ml of the 86 ml pull will have a bunch of nucleated cells (and stem cells). The second 10 ml from the same site will have about half that number and then the third aliquot half that number. All the way up to 86 ml. Hence, in the diagram above you can graphically see the declining number of nucleated and stem cells as the surgeon draws more and more volume from a single site.
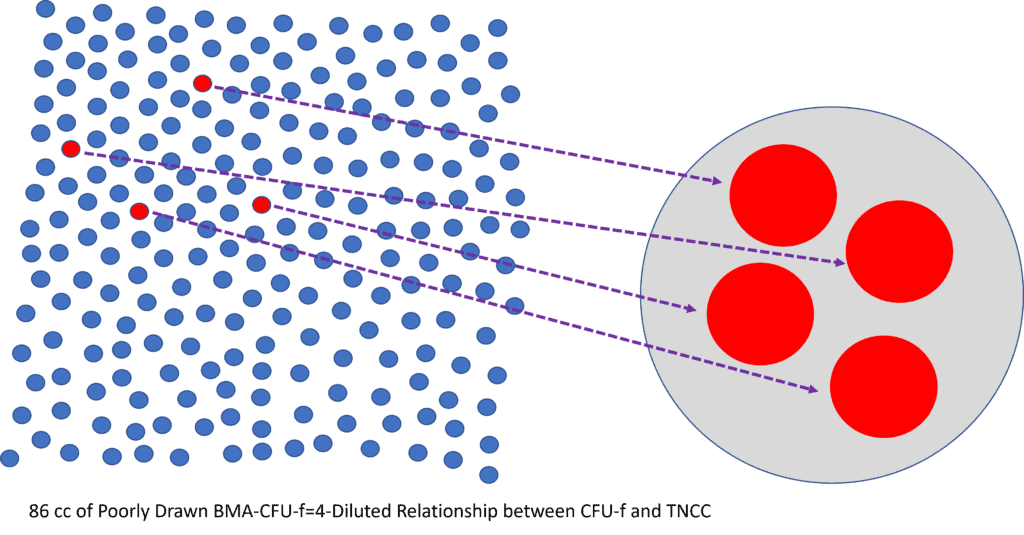
In the illustration above, I took all of those cells from the 86 ml draw and put them closer together so you can get a visual of the number of stem cells in relationship to the total cells (TNCC). Hence, we have a sample that is TNCC and stem cell poor.
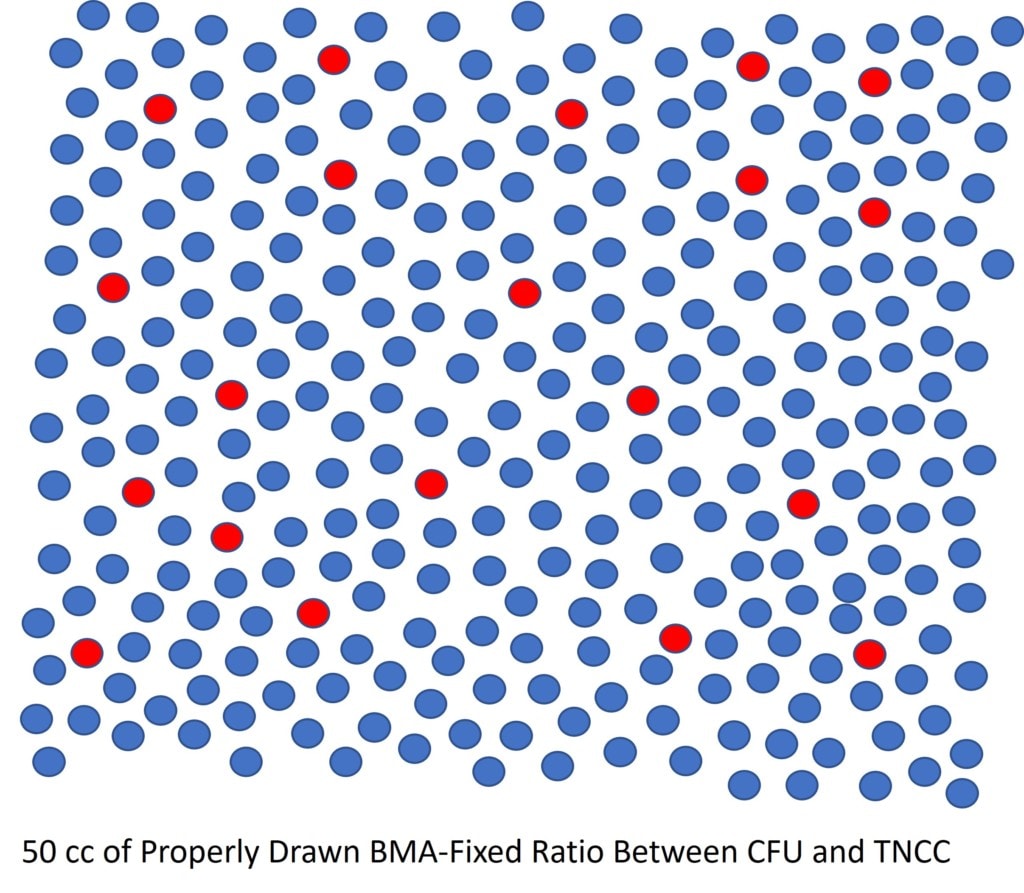
In the diagram above, I modeled what a proper multi-site/low volume draw would produce in 50 ml. First, we have more total cells here in 50 ml than we had in 86 ml. In addition, we have lots more stem cells (red).
Why a Bad BMA Makes this Study Useless
As you can see above, the really bad BMA technique produces few nucleated and stem cells. Hence, that BMA isn’t representative of what’s in the marrow, as only the first 10 ml had a fixed ratio of stem cells to TNCC. Once we pulled the second, third, and subsequent 10 ml aliquots, the dilution by nucleated and stem cell poor peripheral blood dramatically changes our ratio of stem cells to TNCC.
Now had our lazy surgeons decided to perform a proper BMA, they would have kept the number of stem cells to TNCC roughly the same throughout the sample. Meaning lots of stem cells and lots of nucleated cells.
So what happens when we try to link the number of stem cells to the TNCC in the bad BMA sample? That’s now hard to do because we have so seriously diluted the sample with peripheral blood, we’re not measuring any property of the bone marrow in that patient. That’s because we just don’t have that much bone marrow aspirate in that sample. That doesn’t happen when we measure the result of a proper BMA, as what’s in our good BMA sample is representative of what’s in the patient’s bone marrow and the ratio of stem cells to TNCC is preserved.
Summary
The researchers in this paper didn’t actually draw bone marrow aspirate. They drew a tiny amount of bone marrow and mostly blood. We know that for sure by looking at the pitiful TNCC counts reported in the paper. Hence, trying to measure the relationship between the TNCC and stem cells in that sample is foolish.
What is the relationship between the stem cell number and TNCC when you don’t screw up the BMA? When we looked at this relationship, across many samples, the TNCC is directly linked to the number of stem cells (8). Other researchers have found the same thing (9). Hence, the credible data on this topic shows that TNCC is a reasonable surrogate for the stem cell content of a bone marrow sample.
The QC Problem
As you can see above, orthobiologics has a very serious quality control problem. Even experienced surgeon researchers got this one wrong and never noticed this error. The surgeon reviewers at the prestigious journal Arthroscopy also never noticed this blatant problem. Why? None of them were ever educated in how to perform a proper BMA in medical school, residency, or fellowship. Nor was there any QC program in place to make sure that they were getting high cell counts from their BMA. If that had been in place, these surgeons would have adopted a better BMA technique to make sure that each BMA had the max number of cells. They also would have quickly realized that taking bone marrow from the humerus bone, while convenient for the surgeon, is leaving lots of stem cells on the table, since the pelvis has a much higher concentration (4). Why didn’t they just perform a proper BMA at the PSIS of the pelvis, like everyone else that does this work? Again, surgeon convenience trumps science.
All of this is the point of our Regenexx network. No organized network of physicians out there is watching over the shoulder of each doctor performing orthobiologics to make sure these procedures are being done right, other than Regenexx. Meaning, we periodically look at the mean TNCC counts of our network sites to make sure they’re in the correct range. If we find problems, like counts that are way too low, we send someone out to observe the doctor’s BMA technique and re-educate where appropriate.
This brings us to another critical QC problem. Nobody else routinely measures the TNCC of every sample, other than Regenexx. Meaning 99+% of the doctors performing BMC procedures who are outside of Regenxx don’t perform TNCC counts at all. Hence, they have no idea of the dose being delivered to the patient. Hence, even if they had a poor BMA technique, they would never know it.
The upshot? A research paper that used a bad BMA technique can teach us quite a bit about how QC is important in both research studies and in practices that use orthobiologics. As a physician, you need to ask yourself, who is performing QC for me? As a patient, you need to ask yourself if anybody is looking over your doctor’s shoulder to make sure they’re doing it right, as if they’re not, how would you know?
_________________________________________________
References:
(1) Batinić D, Marusić M, Pavletić Z, Bogdanić V, Uzarević B, Nemet D, Labar B. Relationship between differing volumes of bone marrow aspirates and their cellular composition. Bone Marrow Transplant. 1990 Aug;6(2):103-7. PMID: 2207448.
(2) Muschler GF, Boehm C, Easley K. Aspiration to obtain osteoblast progenitor cells from human bone marrow: the influence of aspiration volume. J Bone Joint Surg Am. 1997 Nov;79(11):1699-709. doi: 10.2106/00004623-199711000-00012. Erratum in: J Bone Joint Surg Am 1998 Feb;80(2):302. PMID: 9384430.
(3) Fennema EM, Renard AJ, Leusink A, van Blitterswijk CA, de Boer J. The effect of bone marrow aspiration strategy on the yield and quality of human mesenchymal stem cells. Acta Orthop. 2009 Oct;80(5):618-21. doi: 10.3109/17453670903278241. PMID: 19916699; PMCID: PMC2823327.
(4) Marx RE, Tursun R. A qualitative and quantitative analysis of autologous human multipotent adult stem cells derived from three anatomic areas by marrow aspiration: tibia, anterior ilium, and posterior ilium. Int J Oral Maxillofac Implants. 2013 Sep-Oct;28(5):e290-4. doi: 10.11607/jomi.te10. PMID: 24066346.
(5) Muench LN, Berthold DP, Kia C, Otto A, Cote MP, McCarthy MB, Mazzocca AD, Mehl J. Nucleated Cell Count Has Negligible Predictive Value for the Number of Colony-Forming Units for Connective Tissue Progenitor Cells (Stem Cells) in Bone Marrow Aspirate Harvested From the Proximal Humerus During Arthroscopic Rotator Cuff Repair. Arthroscopy. 2021 Jul;37(7):2043-2052. doi: 10.1016/j.arthro.2021.01.064. Epub 2021 Feb 11. PMID: 33581306.
(6) Centeno CJ, Al-Sayegh H, Bashir J, Goodyear S, Freeman MD. A dose response analysis of a specific bone marrow concentrate treatment protocol for knee osteoarthritis. BMC Musculoskelet Disord. 2015 Sep 18;16:258. doi: 10.1186/s12891-015-0714-z. PMID: 26385099; PMCID: PMC4575428.
(7) Muench, L.N., Kia, C., Otto, A. et al. The effect of a single consecutive volume aspiration on concentrated bone marrow from the proximal humerus for clinical application. BMC Musculoskelet Disord 20, 543 (2019). https://doi.org/10.1186/s12891-019-2924-2
(8) Berger DR, Aune ET, Centeno CJ, Steinmetz NJ. Cryopreserved bone marrow aspirate concentrate as a cell source for the colony-forming unit fibroblast assay. Cytotherapy. 2020 Sep;22(9):486-493. doi: 10.1016/j.jcyt.2020.04.091. Epub 2020 Jun 19. PMID: 32565131.
(9) Mantripragada VP, Boehm C, Bova W, Briskin I, Piuzzi NS, Muschler GF. Patient Age and Cell Concentration Influence Prevalence and Concentration of Progenitors in Bone Marrow Aspirates: An Analysis of 436 Patients. J Bone Joint Surg Am. 2021 Sep 1;103(17):1628-1636. doi: 10.2106/JBJS.20.02055. PMID: 33844657.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
