Infusio: An Infusion for All That Ails You
I recently came across a clinic that uses IV exosomes and other things to help cure or treat a large panoply of diseases. So let’s dive into the cellar medicine aspects of this type of practice through the lens of exosomes. Why exosomes? Because in my opinion, there is no single more abused therapy right now in all of regenerative medicine.
What Is an Exosome?
Exosomes are cellular messengers (1). They’re excreted by all cells in your body as a way of “talking” to each other. What those messages actually say is a concept that’s still in its scientific infancy. However, that hasn’t stopped clinics from advertising stem cell derived exosomes as miracle cures. To help learn more, see my video below:
What Is Infusio?
Infusio is a naturopathic clinic treating a wide variety of serious medical conditions from Lyme disease to knee arthritis to cancer. There seems to be an office in Germany and one in Beverly Hills, but there are some references online to the California clinic closing.
The “Medical” Director of Infusio
Phillip Battiade is listed as the medical director of this clinic. He’s actually an “HP” which is German for “Heilpraktiker” which is a licensed alternative health certificate in Germany. Regrettably, you don’t need any type of formal medical education to get a German Heilpraktiker license, all you need to do is to take a certification exam!
I have complained about naturopaths and their lack of training to be able to perform complex interventional procedures. However, US naturopaths must go through some medical training before being able to practice. Not so for a German Heilpraktiker. This is an article on Philip Battaide or “Dr. Phil”, as he refers to himself.
Exosomes and Infusio
One of the types of treatment that Infusio uses to treat disease is exosomes. Reading the website, I was lead to believe that “Dr.” Battaide was an expert on this topic. Hence I ran a US National Library of Medicine search to find his publications on exosomes and this is what I found:
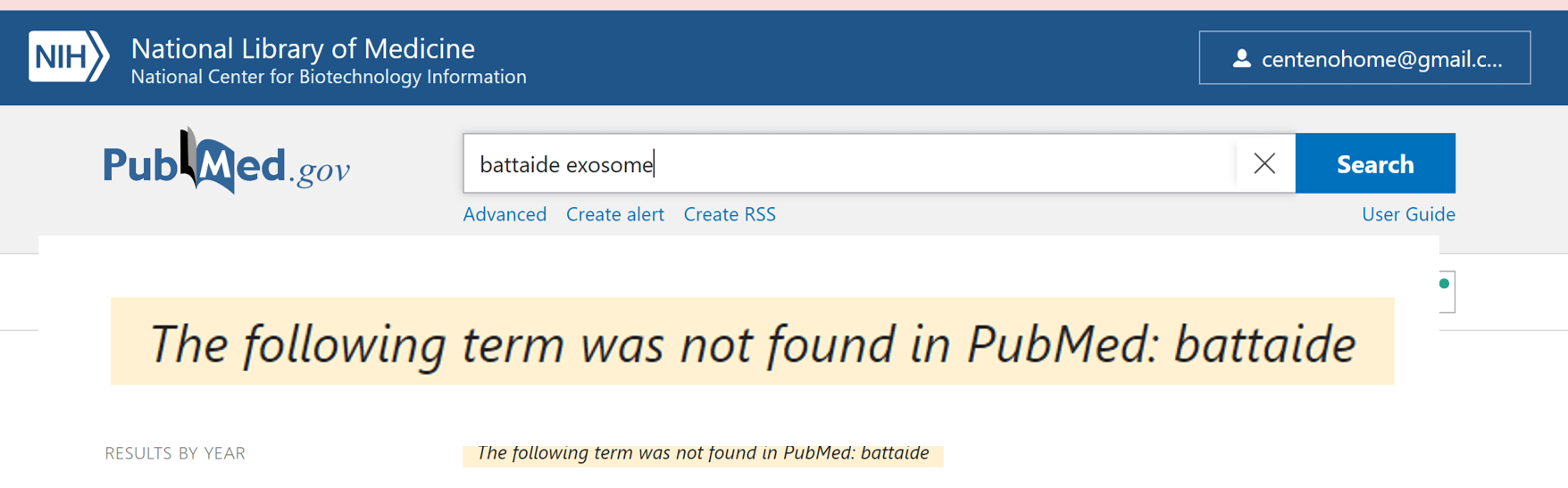
So much for the term “expert”.
Infusio’s Exosome Claims
First, it’s critical to understand that there are hundreds of different types of exosomes that we can identify and likely thousands more we can’t, so identifying which type does which function with any certainty is not possible right now. In fact, even using the term “exosomes” to describe a treatment is like using the term “pill”. We all intuitively know that there can be thousands of different things that can be placed into a pill and that each does something different. So if a pill contains the ingredient “A” you get one effect and if it contains ingredient “B” you get a different effect. Hence, for the Infusio website to say that they use “exosomes” is non-sensical from a medical standpoint. The biggest issue is what type and where is your research on what that does in a human body?
Now let’s vet some claims:
Claim 1:
“Exosomes are extracellular vesicles, or small bubbles, released from cells that act as shuttles for genetic information, proteins and messenger RNA to other cells. This usually happens in response to injuries. Generally speaking, Exosomes carry healthy and lost information and insert this to target cells. Exosomes released by young stem cells have shown to be very powerful in regulating regenerative processes in the body and assisting in rejuvenation.”
Reality: We have no studies published on actual patients showing that exosomes released by young stem cells assist in regeneration and rejuvenation.
Claim 2:
“Are There Any Side Effects?…No long-term negative side effects have been reported.”
Reality: The US FDA put out this warning on exosome products (2):
“The Food and Drug Administration (FDA) is informing the public, especially patients, health care practitioners, and clinics, of multiple recent reports of serious adverse events experienced by patients in Nebraska who were treated with unapproved products marketed as containing exosomes. These reports were brought to the agency’s attention by the Centers for Disease Control and Prevention, among others, and the agencies worked with the Nebraska Department of Health and Human Services. FDA is carefully assessing this situation along with our federal and state partners.
There are currently no FDA-approved exosome products. Certain clinics across the country, including some that manufacture or market violative “stem cell” products, are now also offering exosome products to patients. They deceive patients with unsubstantiated claims about the potential for these products to prevent, treat or cure various diseases or conditions. They may claim that they these products do not fall under the regulatory provisions for drugs and biological products – that is simply untrue. As a general matter, exosomes used to treat diseases and conditions in humans are regulated as drugs and biological products under the Public Health Service Act and the Federal Food Drug and Cosmetic Act and are subject to premarket review and approval requirements.”
Claim 3:
“Are All Exosomes the Same for Every Patient or Can They Be Individualized? And How Would They Be Individualized?
There are different types of exosomes. There is a general standard exosome solution produced from placental derived mesenchymal stem cells. These stem cells are GMP laboratory stem cells, which have been screened and certified.”
Reality:
There is no well studied or clinically accepted “general standard exosome solution” that is produced from placental derived mesenchymal stem cells. In particular, in the FDA warning above, the FDA makes it clear that any clinic offering exosome therapy right now is in violation of US federal law and is selling an unapproved drug product.
The upshot? Infusio isn’t the only clinic out there advertising that exosomes may help cure Lyme disease, aging, chronic inflammation, autoimmune disease, and other degenerative diseases. In fact, there are hundreds of clinics making these claims without any published clinical evidence to support them. As you can see above, we don’t even know yet how to identify the likely thousands of different types of exosomes or what they do. Hence, while exosomes may one day be used clinically, right now, based on the lack of clinical evidence that Infusio’s or any other product do anything good for patients, this technology, in my opinion, is not ready for clinical use.
__________________________________________________
(1) Gurunathan S, Kang MH, Jeyaraj M, Qasim M, Kim JH. Review of the Isolation, Characterization, Biological Function, and Multifarious Therapeutic Approaches of Exosomes. Cells. 2019;8(4):307. Published 2019 Apr 3. doi:10.3390/cells8040307
(2) US Food and Drug Administration. Public Safety Notification on Exosome Products. https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/public-safety-notification-exosome-products Accessed 6/2/20.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
