Stem Cell Injection Safety Research
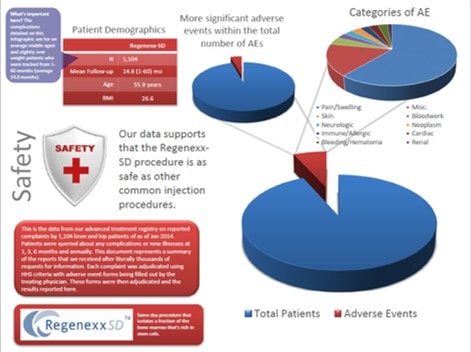
Safety has always been a big concern of ours. As a result, we have published more data on stem cell safety in the peer reviewed medical research for orthopedic applications than any other group world-wide. For example, our n=339 stem cell safety paper published in 2011 was named the best of it’s class by independent European researchers last year. In keeping with that trend, above is a patient infographic of safety data that will be included in an upcoming research paper. This will focus on the safety and efficacy of 1,104 knee and hip arthritis patients treated with the Regenexx-SD same day stem cell procedure. While the document linked above (click on the thumbnail to see the bigger PDF) is only two pages, it represents a massive amount of work over 5 years by many people. To collect this safety data we had to build a massive treatment registry infrastructure costing hundreds of thousands of dollars; quite a bit of money for a small clinic. We had to hire staff to track all of these patients electronically and on the phone. In addition, thousands upon thousands of e-mails and phone calls were made to elicit any complaints. Finally, hundreds of hours of physician time then went into investigating and classifying these possible side effects. Nobody else involved in using stem cells as part of daily practice to treat orthopedic conditions that we know of takes this so seriously, and nobody routinely reports this type of safety data in this level of detail on a regular basis. What did all of this stem cell injection safety research show? Based on our analysis of this treatment registry data, the Regenexx-SD procedure is about as safe as any typical injection procedure, which is consistent with what we see every day in the clinic.

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.