Stem Cell Treatment for Torn Rotator Cuff : Our New Research
We’ve been offering patients stem cell treatment for torn rotator cuff tendons for almost 15 years. In fact, we were the first clinic on earth to use this as a primary treatment for rotator cuff tears. Along the way, we’ve published some research on this treatment and learned quite a bit. Let’s dive into this topic.
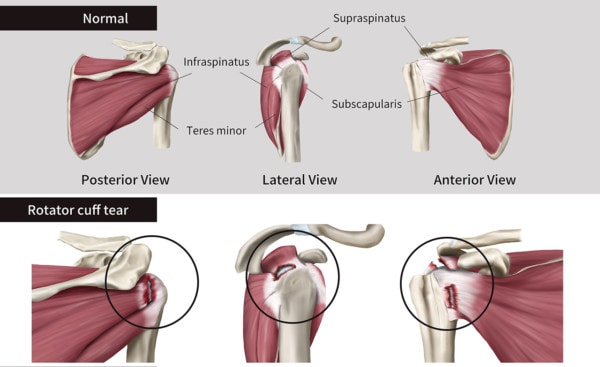
What Is the Rotator Cuff?
Chu KyungMin/Shutterstock
This is a group of muscles that surround the shoulder joint and both stabilize and move it. These tendons can become torn through trauma or wear and tear. When they do, the tear can be partial or complete. If the latter, the fibers can still be intact or they can pull back like a rubber band leading to a massive tear. Here we’re focusing on partial and complete non-retracted tears which make up about 2/3rds of what’s called a rotator cuff tear.
Is There an Alternative to Rotator Cuff Surgery?
Rotator cuff surgery normally involves surgically sewing together the torn parts, but this often comes with major downtime in an immobilizer pillow. In addition, despite this invasive procedure, an analysis of 11 different research studies concluded that surgery didn’t work any better than physical therapy at restoring shoulder function (4). So are there alternatives to this type of surgery? How about Stem cell treatment for torn rotator cuff tendons?
Two of the most promising treatments to replace rotator cuff surgery are platelet-rich plasma (PRP) and stem cells. PRP is created by concentrating blood platelets using a centrifuge and then, in this case, injecting that using ultrasound guidance precisely into the tears. Based on existing research studies this injection therapy seems to work in patients with partial tears (5-7).
The other type of treatment that may be able to heal the tear is stem cell treatment for torn rotator cuff tendons. Here, the doctor takes a bone marrow sample from the back of the hip and then uses a centrifuge to isolate the stem cell fraction. That’s then precisely injected into the tear. To see what that procedure looks like, check out my video below:
Does the Research Show that Stem Cell Treatment Works?
Regenexx has a long history of publishing research. I got a kick out of this Google search I did the other day for an upcoming lecture that showed that one of our first papers on the use of stem cells in orthopedics was the most cited paper listed. This means that it has the most research papers that came after it that have referenced this work. That’s a great honor (see below).
We first published on the use of a same-day stem cell procedure to treat shoulder arthritis and rotator cuff tears way back in 2015 (1). We also included a safety analysis on a larger group of treated patients who had stem cell treatment for torn rotator cuff tendons in a 2016 paper (2). This week, our group published the mid-term analysis of a multi-year shoulder rotator cuff randomized controlled trial. The paper demonstrated that the precise injection of high-dose bone marrow concentrate into the damaged rotator cuff tendon using ultrasound guidance helped many patients recover without the need for surgery (3). Others have also published research on the use of bone marrow concentrate (a same-day stem cell procedure) to treat rotator cuff tears either through injections or with surgery (8-10).
How Much Does Shoulder Stem Cell therapy Cost?
First, realize that the type of stem cell treatment we have discussed here uses the patient’s own bone marrow and NOT umbilical cord tissue. While many clinics offer stem cell treatment for torn rotator cuff tendons using umbilical cord “stem cells”, several studies have shown that this is a bait and switch fraud as the products being used have no viable stem cells (11-13). Hence, if you get offered umbilical cord or amniotic “stem cells” at any price, that’s a scam. Check out my video below on the subject:
Second, there are some insurance carriers through large employers who have begun to cover these bone marrow stem cell injection procedures. Finally, the average cash cost can be as low as $3-4,000 to as high as $12-15,000. The reputable clinics who are doing good work and who use imaging guidance and high dose stem cell harvest and processing techniques tend to be in the 5-7K range. Generally, clinics that are charging less are cutting corners, and clinics charging much more are overpriced.
The upshot? Stem cell treatment for torn rotator cuff tendons is available and the research is continuing to support that it likely works. However, be very careful about getting taken, as most offerings are either low quality or outright fiction. Hence, do your homework!
____________________________
References:
(1) Centeno CJ, Al-Sayegh H, Bashir J, Goodyear S, Freeman MD. A prospective multi-site registry study of a specific protocol of autologous bone marrow concentrate for the treatment of shoulder rotator cuff tears and osteoarthritis. J Pain Res. 2015;8:269–276. Published 2015 Jun 5. doi:10.2147/JPR.S80872
(2) Centeno CJ, Al-Sayegh H, Freeman MD, Smith J, Murrell WD, Bubnov R. A multi-center analysis of adverse events among two thousand, three hundred and seventy two adult patients undergoing adult autologous stem cell therapy for orthopaedic conditions. Int Orthop. 2016 Aug;40(8):1755-1765. doi: 10.1007/s00264-016-3162-y.
(3) Centeno CJ, Fausel Z, Stemper I, Azuiike U, Dodson E. A Randomized Controlled Trial of the Treatment of Rotator Cuff Tears with Bone Marrow Concentrate and Platelet Products Compared to Exercise Therapy: A Midterm Analysis. Stem Cells International. 2020 Jan; 2020. https://doi.org/10.1155/2020/5962354
(4) Centeno CJ, Al-Sayegh H, Bashir J, Goodyear S, Freeman MD. A prospective multi-site registry study of a specific protocol of autologous bone marrow concentrate for the treatment of shoulder rotator cuff tears and osteoarthritis. J Pain Res. 2015;8:269–276. Published 2015 Jun 5. doi:10.2147/JPR.S80872
(5) Ilhanli I, Guder N, Gul M. Platelet-Rich Plasma Treatment With Physical Therapy in Chronic Partial Supraspinatus Tears. Iran Red Crescent Med J. 2015;17(9):e23732. Published 2015 Sep 28. doi: 10.5812/ircmj.23732
(6) Cai YU, Sun Z, Liao B, Song Z, Xiao T, Zhu P. Sodium Hyaluronate and Platelet-Rich Plasma for Partial-Thickness Rotator Cuff Tears. Med Sci Sports Exerc. 2019;51(2):227–233. doi: 10.1249/MSS.0000000000001781
(7) Rha DW, Park GY, Kim YK, Kim MT, Lee SC. Comparison of the therapeutic effects of ultrasound-guided platelet-rich plasma injection and dry needling in rotator cuff disease: a randomized controlled trial. Clin Rehabil. 2013 Feb;27(2):113-22. doi: 10.1177/0269215512448388.
(8) Kim SJ, Kim EK, Kim SJ, Song DH. Effects of bone marrow aspirate concentrate and platelet-rich plasma on patients with partial tear of the rotator cuff tendon. J Orthop Surg Res. 2018;13(1):1. Published 2018 Jan 3. doi:10.1186/s13018-017-0693-x
(9) Kim SJ, Song DH, Park JW, Park S, Kim SJ. Effect of Bone Marrow Aspirate Concentrate-Platelet-Rich Plasma on Tendon-Derived Stem Cells and Rotator Cuff Tendon Tear. Cell Transplant. 2017;26(5):867–878. doi:10.3727/096368917X694705
(10) Hernigou P, Flouzat Lachaniette CH, Delambre J, Zilber S, Duffiet P, Chevallier N, Rouard H. Biologic augmentation of rotator cuff repair with mesenchymal stem cells during arthroscopy improves healing and prevents further tears: a case-controlled study. Int Orthop. 2014 Sep;38(9):1811-8. doi: 10.1007/s00264-014-2391-1.
(11) Berger DR, Lyons NF, Steinmetz NJ. In Vitro Evaluation of Injectable, Placental Tissue-Derived Products for Interventional Orthopedics. Interventional Orthopedics Foundation Annual Meeting. Denver, 2015. https://interventionalorthopedics.org/wp-content/uploads/2017/08/AmnioProducts-Poster.pdf
(12) Becktell L, Matuska A, Hon S, Delco ML, Cole B, Fortier LA. Proteomic analysis and cell viability of nine amnion-derived biologics. Orthopedic Research Society Annual Meeting, New Orleans, 2018. https://app.box.com/s/vcx7uw17gupg9ki06i57lno1tbjmzwaf
(13) Panero, A. J., Hirahara, A. M., Andersen, W. J., Rothenberg, J., Fierro, F. Are Amniotic Fluid Products Stem Cell Therapies? A Study of Amniotic Fluid Preparations for Mesenchymal Stem Cells With Bone Marrow Comparison. The American Journal of Sports Medicine, 47(5), 1230–1235. https://doi.org/10.1177/0363546519829034

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
