The Regenexx Difference-Cell Viability or are the stem cells you’re getting dead on arrival?
Cell viability means the health of the cells. For stem cell therapies, it’s critical that the stem cells you’re receiving are viable (healthy). While this may seem like a simple thing, it’s really not. Stem cells are fragile cells that don’t do well outside of the body and small things like mechanical trauma from collection or exposure to certain drugs can have a big negative effect on their viability. As an example, above I’ve included a series of microscope images from our lab. In this experiment, we simply exposed mesenchymal stem cells in culture to commonly used anesthetics. Note that the two most commonly used anesthetics (lidocaine and marcaine) killed cells dead. What’s more surprising is that they killed stem cells as very low concentrations (well below the levels used by a doctor in any common medical procedure). Physicians recently learned how toxic these anesthetics were the hard way-shoulder surgery pain pumps delivering a continuous infusion of local anesthetics resulted in destroying all cartilage cells in the joint. How might this anesthetic effect on stem cells impact your planned stem cell treatment? As one example, the most commonly used collection methods for stem cells often involve using these common anesthetics. Liposuction is illustrative-here massive amounts of local anesthetics are injected directly into the tissue containing stem cells that will be harvested. Placing stem cells back into a joint is accomplished in many clinics with injection of anesthetics, which will hurt the cells.
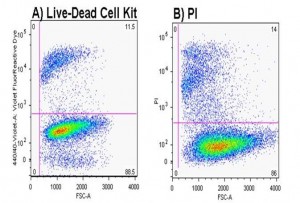
Other aspects of cell viability are important to note as well. Cells can be damaged by collection methods or in the case of bone marrow, the doctor may not be taking stem cells at all, but instead peripheral blood that doesn’t contain the type of stem cells sought. Mechanical trauma means that the method used to collect cells or re-inject cells may physically hurt or kill cells, reducing the number that can help repair tissue. Above is an example of how we have used flow cytometry (a complex piece of equipment capable of looking at markers on each cell) to check this viability. Without this capability to check stem cell viability, there’s no way for the patient to be sure that whatever method the doctor is using to collect stem cells actually results in live stem cells going back into the patient. Unlike other physicians offering stem cell treatments, the Centeno-Schultz Clinic has a state of the art cell biology lab as part of the practice. This allows us to confirm that the methods we use to harvest stem cells (bone marrow, fat, or blood) actually produce viable cells capable of growing in culture.
What questions should you ask the stem cell clinic?
1. Do you have an on-site lab or have you sent your stem cells to an off-site lab for verification that the methods you use (not those that were taught, but the ones that you use) actually produce viable stem cells? Can I see that verification report from cells collected at your facility?
2. Do you allow the stem cells to come in contact with local anesthetics?
3. If collecting a marrow aspirate to obtain stem cells, does the doctor use imaging guidance to ensure that he’s in the right spot containing bone marrow stem cells or does he perform the procedure blind?
4. If the cells are transported to another facility for processing or kept for any time in a tube before being placed back into the patient, what tests have been performed in your facility on cell viability (see question 1)?
5. How much bone marrow aspirate do you take from each spot? (If over about 10-20 ml from each spot, our data and that published by others show that marrow stem cells aren’t being harvested, just a bunch of blood that doesn’t contain the type of bone marrow stem cell being targeted)

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.

