A Big Fat Blow for the Stem Cell Literature: Major Paper Withdrawn
This past year, I was actually excited to add fat stem cells to my big annual review of the orthopedic stem cell literature. While a few small papers had been published, a big part of that literature was a huge data set by Michalek out of Eastern Europe, which purported to show that fat stem cells used for knee arthritis worked great. While I was a critic of the paper because the results seemed too good to be true, I felt it needed to be included in the discussion over bone marrow versus fat. However, this past week I learned that the paper had been withdrawn, which decimates the fat stem cell research literature.
The Back Story on Bone Marrow vs. Fat
While we have chiropractic-clinic stem cell seminar scams popping up all over, promising to regrow new knees with zombie (living-dead) amniotic tissue, real doctors who do this work have been arguing back and forth for the last decade about whether bone marrow or fat was better for orthopedic purposes. The fat-stem-cell crowd has argued that fat has more stem cells (please see video below as this is largely untrue) and that a liposuction is less invasive than a bone marrow aspirate (again untrue). Having said that, there has been a paucity of clinical studies on fat stem cells to date compared to much more on the use of bone marrow concentrate in orthopedics.
The Study That Seemed to Alter the Fat-Stem-Cell-Research Calculus
In 2015, a massive data set of knee and hip arthritis patients treated with both types of fat stem cell therapies (stromal vascular fraction, or SVF, which is illegal in the U.S.) and a fat graft was published by Michalek. This fat-stem-cell paper looked a bit strange to me, so I pointed out a few problems with the research methods. What was wrong?
First, the follow-up rate was ridiculously high. In any study, certain patients will be lost to follow-up, meaning that after a time, they fail to respond. This is unavoidable, but if the study is small, you can usually minimize the number of patients that don’t respond to your questionnaires. However, once the study gets to more than 100 patients, this can be tough. If the study gets to several hundred patients, it’s almost impossible. This study had more than a thousand patients, and the follow-up rate was much higher than you would expect. In fact, without involving the local police and SWAT unit to hunt people down who failed to fill out their outcome forms, it seemed suspiciously high.
Second, the results seemed too good. The study reported that a very high number of patients had fantastic results. In fact, that 91% of patients were classified as having greater than 50% improvement at one year after the injection. These numbers were much higher than any other publication on any published stem cell procedure. When digging into the paper, it wasn’t hard to see how this sleight of hand was accomplished. The physician authors got to make up 60% of the final outcome, while only 40% of the input came from the patient.
The Study Is Withdrawn
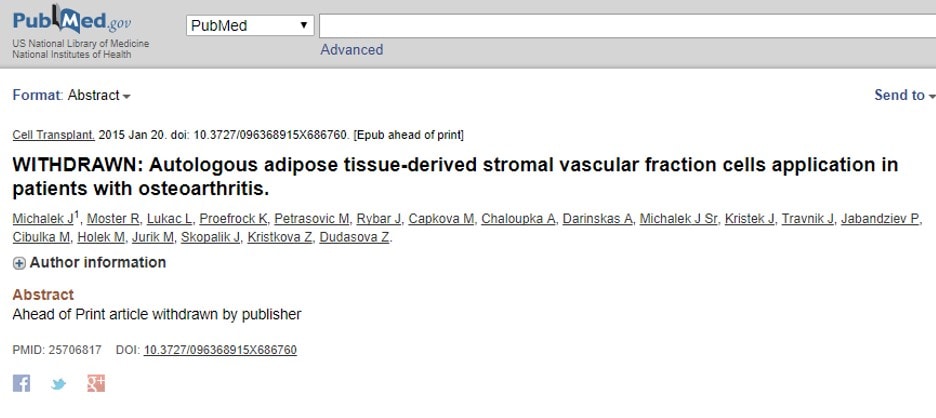
This past week, I went to look for the study to cite it in another work, and I found this:
Was the paper pulled from the journal for the problems I had identified, or was it something else? It’s likely we may never know for sure.
What I can say is that the withdrawal of this paper from the fat-stem-cell literature punches a huge gaping hole in the research base. For example, my 2017 version of the summary of fat-stem-cell research had 10,018 patients who had their results reported for any stem cell type to treat orthopedic injuries. Of that, 8,533 were patients treated with bone marrow stem cells. A whopping 1,128 of the 1,485 patients treated with other cell types were from the Michalek study. Subtract those patients, and the totality of non-bone-marrow-treated patients falls to 357 compared to 8,533 for bone marrow. In summary, take out the Michalek study, and the research on stem cells other than bone marrow used to treat orthopedic problems is almost insignificant.
The upshot? The withdrawal of the Michalek paper is a huge blow for the research on fat stem cells in orthopedics. Hopefully, more research will be published quickly to bridge the huge chasm of research support for fat stem cells compared to bone marrow!
9/12/17-I was contacted by the author of the above paper and it was republished here after being withdrawn by the editor of the initial publisher: http://www.oatext.com/pdf/GOS-3-163.pdf . While the author and I disagree on outcome measurement, I do feel that this paper is a key one for the orthopedic adipose stem cell literature.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.