Is R3 Stem Cell Really an Award Winner?

R3 is a marketing company run by a defrocked orthopedic spine surgeon that has gained lots of attention for flaunting FDA regulations and claims about its private-labeled stem cell products that can’t be verified. Not to be dissuaded, they have now created a host of artificial awards and press releases or what I called the “circle of self-promotion”. Let’s dig in.
What is R3 Stem Cell?
Prior to the beginning of the stem cell wild west, in 2009, an orthopedic spine surgeon named David Greene lost his medical license. This was for multiple botched spine surgeries resulting in patient harm. Here is the order from the Arizona medical board:

After that, he reinvented himself in healthcare marketing and in 2013 formed R3. This was essentially a marketing company that rebranded other people’s birth tissue products as “stem cells”.
In October of 2020, I featured R3 on the blog for fraudulently claiming that its products could be billed to Medicare to treat orthopedic problems. In addition, a naturopath who was teaching “stem cell” courses for R3 was performing liposuction in one of their videos, which was outside the scope of practice for naturopaths in places where the course was taught.
R3 and the FDA
In May 2019, R3 got a letter from the FDA because it was promoting stem cell therapies for numerous diseases or conditions such as dementia, Parkinson’s disease, and ALS. That was accompanied by a press release from the FDA:
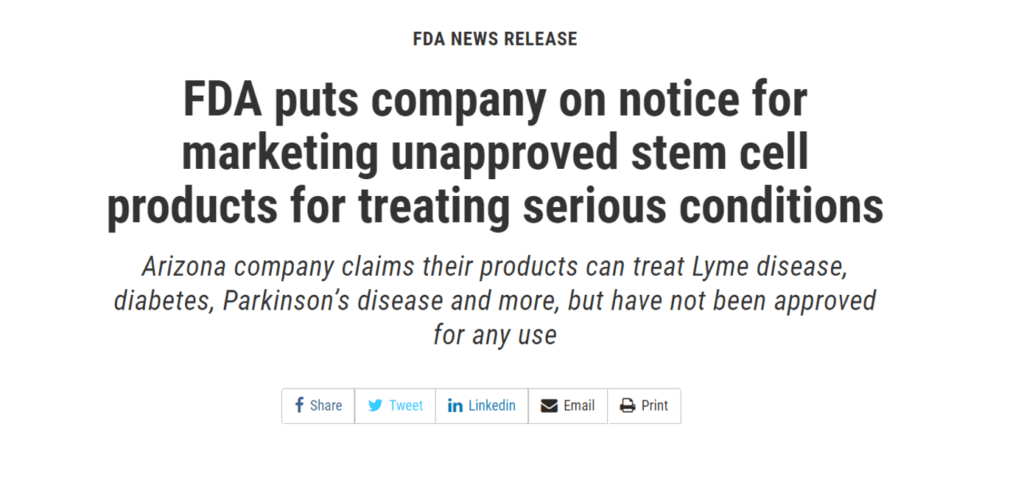
R3 stem cell was also featured by ProPublica in its piece on birth tissue profiteers:
The Newest Press Release

I recently came across the above press release from R3. First, notice that in the grey box in the lower right-hand corner of the image, the contact is David Greene and the name of the company is “US Lead Network”. Again, as I said, R3 is a marketing company. It doesn’t make any products nor perform any research, it just private labels the products of others, holds courses, and connects its clinic buyers with patient leads.
What first astounded me by the press release was the complete disregard for the prior FDA letter in statements like this:
“The founder and CEO of R3 Stem Cell, Dr. David Greene aims at making the planet earth fully free from diseases by ensuring the availability of fully organic and risk-free treatment to every human. The progressive research and studies in Regenerative Medicine or RM have discovered numerous types of stem cells that all don’t go equally in favor of humans given their pros and cons. R3 Stem Cell offers only FDA-regulated RM procedures including Amniotic Fluid Stem Cells (AFSCs), Umbilical Cord Stem Cell (UCSCs), Exosomes, and PRP Therapies that perform natural rejuvenation of cells and tissues in the body. With this technological treatment, you are saved from any type of side-effects or surgeries.”
IMHO, if you could design a statement that was non-FDA compliant and violate the letter that the agency had already sent you as well as multiple other FDA statements and warnings, this would be what you would write. In fact, claiming that you’re selling amniotic and umbilical cord stem cells and is not only fraudulent based on the existing research, but sure to cause problems (1-3).
If you’re new to the whole fake amniotic and umbilical cord stem cell game being played by many companies including R3, then watch my two videos below. If you know this stuff, skip ahead to read about the phony awards in the press release:
Phony Awards?
The press release outlines various “news” pieces that seem favorable to R3. Take for example:
“2 NEWS
This article talks about R3 Stem Cell winning the 2021 Most Outstanding Regenerative Medicine Company Award. The award is not a mere appreciation but recognition and acknowledgment of the credibility of stem cell treatments offered at R3 Stem Cell.”
So did Channel 2 news actually cover this coveted award? Nope. This is another press release that the Channel 2 news site automatically picked up and posted on their web-page. What was the award? The “2021 Most Outstanding Regenerative Medicine Company”! The problem, that award was given by a company that sells pay-for-play awards called Corporate Vision.
Cision PR Web
The press release goes on to relay, “The renowned and credible digital platform Cision PRWeb talks about R3 Stem Cell offering regenerative medicine procedures for Stroke in Mexico.” Who is Cision PRWeb? A press release hosting site. That means that you write a press release and pay them to host it. So PR Cision never talked about R3 at all, the company merely wrote a release and paid PR Cision to post it.
10 best Healthcare Companies of 2020?
The R3 press release goes on to highlight “EINPRESSWIRE”:
“This international press release talks about R3 Stem Cell headlining the 10 best healthcare companies of the year 2020. Winning awards, ranking top of the list, and ensuring promising stem cell treatments wipes out all the questions raised upon the credibility of R3 Stem Cell.”
The statement, “wipes out all the questions raised upon the credibility of R3 Stem Cell” is interesting. This “news story” is yet another press release from R3 hosted on a pay-for-play press release site. It was given by a company calling itself “CXO Fortune”. A simple Google search of that unique name finds a Linkedin page of a marketing group in Billings, Montana. They seem to run a magazine that features companies and awards:

Notice that the red arrow above points to a drop-down menu item that says “Pay Now”. That seems to make sense because we also find this:

This is a testimonial from none other than David Greene who is thanking them for their business relationship and how his company was presented. So why is the guy they gave an award to producing a testimonial for their magazine?
The Ring of Self-Promotion
So to summarize, we have a marketing company run by an ex-orthopedic spine surgeon who had his medical license revoked. He reinvents himself as a lead generator marketing company and then as someone selling stem cells to clinics. He gets sideways with the FDA who calls out that he is violating their regulations by claiming to sell stem cells, which based on the published and presented literature is itself a fraud because these products contain no live nor viable stem cells. However, he then puts out a subsequent press release continuing to claim to sell stem cells to make “the planet earth fully free from diseases”, which IMHO again violates FDA regulations. He then gives himself a string of what appear to be phony awards. Again, you can’t make this stuff up!
The upshot? We’ve seen this before. For some reason, stem cell marketing groups love to give themselves fake awards or ones that they invent. However, on this one, even I was surprised and at this point, not much surprises me.
___________________________________
References:
(1) Berger D, Lyons N, Steinmetz, N. In Vitro Evaluation of Injectable, Placental Tissue-Derived Products for Interventional Orthopedics. Interventional Orthopedics Foundation Annual Meeting. Denver, 2015. https://interventionalorthopedics.org/wp-content/uploads/2017/08/AmnioProducts-Poster.pdf
(2) Becktell L, Matuska A, Hon S, Delco M, Cole B, Fortier L. Proteomic analysis and cell viability of nine amnion-derived biologics. Orthopedic Research Society Annual Meeting, New Orleans, 2018. https://app.box.com/s/vcx7uw17gupg9ki06i57lno1tbjmzwaf
(3) Panero, A, Hirahara, A., Andersen, W, Rothenberg J, Fierro, F. Are Amniotic Fluid Products Stem Cell Therapies? A Study of Amniotic Fluid Preparations for Mesenchymal Stem Cells With Bone Marrow Comparison. The American Journal of Sports Medicine, 2019 47(5), 1230–1235. https://doi.org/10.1177/0363546519829034

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
