New Study On Stem Cells and Knee Arthritis
There has recently been published a new study on stem cells and knee arthritis. We’ve published quite a bit on the use of mesenchymal stem cells for various forms of arthritis. We just had a hand arthritis paper accepted for publication last week and also have published a large comparison trial in knee arthritis. We will be digging into our new CRO quality database this summer and publishing more research. One of the hallmarks of a therapy that works is when others try the same thing and also publish positive results. This small study is from a group in Spain and it too shows efficacy for the use of stem cells and knee arthritis. Their report of improvement is similar to what we published in a larger study and they also reported improvement in cartilage signal on T2 MRI mapping in 11/12 patients (which we’ve also shown and featured on this site). Below is the abstract from PubMed:
Treatment of Knee Osteoarthritis with Autologous Mesenchymal Stem Cells: A Pilot Study
Orozco, Lluis; Munar, Anna; Soler, Robert; Alberca, Mercedes; Soler, Francesc; Huguet, Marina; Sentís, Joan; Sánchez, Ana; García-Sancho, Javier
Transplantation . POST AUTHOR CORRECTIONS, 15 May 2013
doi: 10.1097/TP.0b013e318291a2da
Abstract
Background: Osteoarthritis is the most prevalent joint disease and a frequent cause of joint pain, functional loss, and disability. Osteoarthritis often becomes chronic, and conventional treatments have demonstrated only modest clinical benefits without lesion reversal. Cell-based therapies have shown encouraging results in both animal studies and a few human case reports. We designed a pilot study to assess the feasibility and safety of osteoarthritis treatment with mesenchymal stromal cells (MSCs) in humans and to obtain early efficacy information for this treatment.
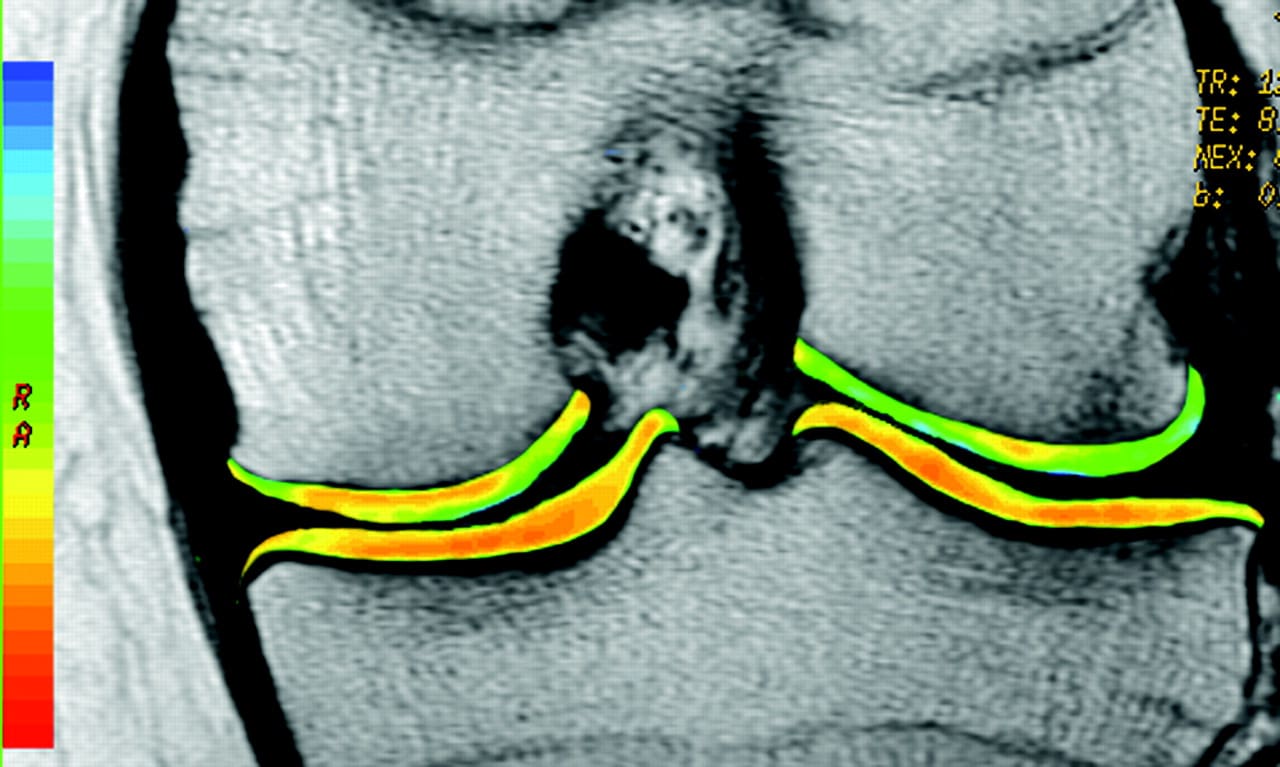
Methods: Twelve patients with chronic knee pain unresponsive to conservative treatments and radiologic evidence of osteoarthritis were treated with autologous expanded bone marrow MSCs by intra-articular injection (40×106 cells). Clinical outcomes were followed for 1 year and included evaluations of pain, disability, and quality of life. Articular cartilage quality was assessed by quantitative magnetic resonance imaging T2 mapping.
Results: Feasibility and safety were confirmed, and strong indications of clinical efficacy were identified. Patients exhibited rapid and progressive improvement of algofunctional indices that approached 65% to 78% by 1 year. This outcome compares favorably with the results of conventional treatments. Additionally, quantification of cartilage quality by T2 relaxation measurements demonstrated a highly significant decrease of poor cartilage areas (on average, 27%), with improvement of cartilage quality in 11 of the 12 patients.
Conclusions: MSC therapy may be a valid alternative treatment for chronic knee osteoarthritis. The intervention is simple, does not require hospitalization or surgery, provides pain relief, and significantly improves cartilage quality.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.