Does Radiographic Contrast Harm Stem Cells?
We’ve been working hard at the task of perfecting orthopedic stem cell treatments for a very long time. Way back when, a decade ago, we ran a small experiment that showed us that we needed to use less radiographic contrast when injecting stem cells. Looks like the rest of the world just caught up with a new study that just demonstrated the same thing. So let’s review this new research and what lessons patients and doctors can learn from this new study and our old in-house research.
What Is Radiographic Contrast?
X-rays only show bones really well, and oftentimes soft tissue, like cartilage, muscles, ligaments, and tendons are invisible. Doctors will often inject a radiographic contrast that can be seen on X-ray to ensure they’re in the joint space. This is the standard for interventional spine and radiology. These contrast agents used to be heavily iodine based and could cause allergic reactions. However, changes were made about 20 years ago, and now newer contrast drugs cause fewer reactions. The most common of these is called Omnipaque (iohexol).
Our Lab Capabilities Are Second to None
We’ve always performed various lab experiments on our own. For example, way back when, we also did the basic science research into whether common local anesthetics were safe to use around stem cells (they’re not). We eventually redid that study and published it. Recently, we also tested PRP dose versus tendon cells in the lab, finding that higher doses were better for older cells. How can we do this work? Unlike other outfits that claim to be “institutes” or “networks,” we actually have a university-level lab at our Colorado HQ. To take a quick tour of that facility, see below:
Iohexol and Stem Cells
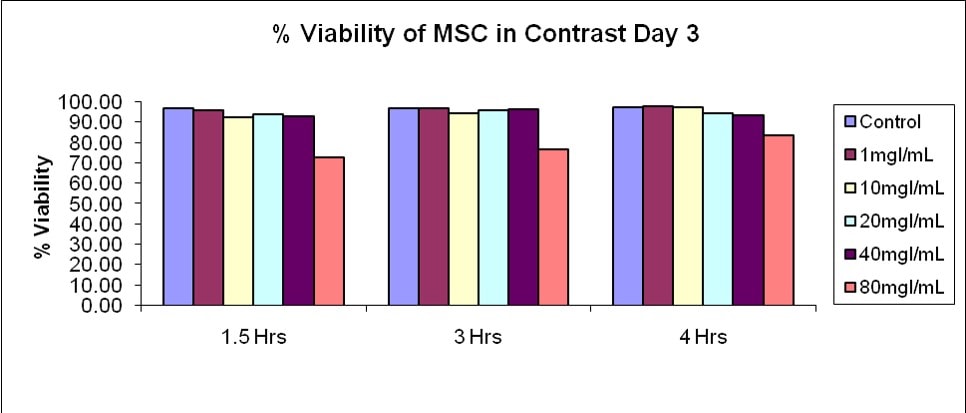
About a decade ago, in 2008, we had been injecting stem cells for three years as part of an IRB-approved study and were just beginning to treat patients. We were one of the only practices on earth doing this work, and there was really no one else in the U.S. who was performing orthopedic stem cell injections. The question came up that since we were often using Omnipaque as a contrast injection with real-time X-rays (fluoroscopy), how would this drug impact the cells? As a result, we did our own experiment. Those results are below:
What did we see? As the concentration of the iohexol in the Omnipaque increased, for the most part, the stem cells did fine. However, as the concentration got higher, there was a point where stem cells began to tank. This was somewhere between 40–80 mg/ml.
What was the take home? For us, this meant that we should either be using very little contrast or we should be diluting it when we were also injecting stem cells. From a practical standpoint, how much is safe? Let’s do the math.
For a knee, the average amount of fluid that already lives there is about 2–5 ml. Let’s call it 3 ml. If we’re injecting 5 cc of stem cells and PRP, then this is 8 ml total fluid in the joint. If we’re using 1/2 ml of Omnipaque 240 contrast, that’s 8.5 ml in the knee. This has 518 mg/ml of iohexol. If we divide that by 2, we have 259 mg injected into the joint. Then if we divide that by 8.5 ml, we get about 30 mg of iohexol, which is within our safe zone above.
What Did the New Study Find?
The new research looked at a few different types of stem cells as well as contrast agents, but to compare to our old in-house research, we’ll focus on iohexol (Omnipaque) and bone marrow mesenchymal stem cells (MSCs). They used Omnipaque 240 and diluted it from 1:1 all the way down to 1:10. They saw that awful things were happening to MSCs at a 1:1 dilution, but that’s also a whopping 259 mg/ml! How much contrast would you need to inject in a joint with 5 ml of stem cells and 3 ml of joint fluid to get there? You would need to inject 8 ml of contrast. Given that most physicians would never exceed 1–2 ml in a joint, that’s not a realistic medical event!
One of the big differences in the new study and our old one was that they exposed the cells “overnight.” This is a severe weakness of this new study as they don’t list the exact time of exposure. A colleague contacted the authors and they relayed that this time was 24 hours. As you can see above, we used 1.5, 3, and 4 hours. This difference in exposure time is HUGE.
How important is exposure time in medicine? As an example, a flame is hot enough to burn flesh. However, if you pass your hand quickly through a flame, nothing happens. Increase the exposure time and you have issues.
So which exposure time is physiologically correct or way off? That all depends on how long iohexol sticks around in the body. A half-life of a drug is the time it takes the body to get rid of half of the drug.
When injected IV, the half-life of iohexol (cleared by the kidneys) is 121 minutes, or 2 hours. Based on the discussion from this study, which references how different drugs are cleared from the knee joint, due to its highly vascular and permeable synovial lining, it’s likely that iohexol has a half-life of hours. So if we use the data we have and conservatively estimate a half-life of 4 hours, only one-half of the dose would be left after that time.
A decade ago we ran our own experiments to determine how long we could observe contrast in the joint. From our observations (i.e., taking serial X-rays), to determine how long the contrast could still be seen, this data also demonstrated that the contrast agent is rapidly cleared. For example, within 1–2 hours, oftentimes much of the contrast wouldn’t be found. Hence, since drugs don’t generally break down in culture, the new study has likely WAY OVEREXPOSED the cells in an unrealistic way that doesn’t mimic what would happen in the body.
At what dose did the cells run into problems in the new study? Omnipaque 240 contains 518 mg/ml of iohexol. Their 1:10 dilution was similar (but slightly higher at 47 mg) to our 40 mg/ml condition. They saw no significant changes in MSC proliferation for iohexol until 1:7 (which is 65 mg/ml). We saw that in our 80 mg/ml condition. Hence, their data roughly matches ours, despite this overexposure. Some of the other changes they observed in cells are likely tied to this huge difference in the time the drug had to act on the cells.
Final Recommendations on Contrast Use with Stem Cells?
If you use Omnipaque 240 or 300, then you have to go light on its use around stem cells. Meaning that, in the average knee, you can’t exceed 1/2 to 1 ml of contrast. You can also dilute this 50/50 with normal saline and inject twice as much or use less-concentrated versions of the product (i.e., Omnipaque 140). If the knee has more native fluid or if you inject higher volumes of a stem cell injectate, you can inject more contrast.
The upshot? We did this study a decade ago, so it’s good to see that the rest of the stem cell world is finally catching up! In the meantime, make sure your doctor knows about all of these little things that matter! If you’re a doctor, then take it easy on the contrast!

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.