Regenexx Reviews: Do Orthobiologics Always Work?

©Regenexx
On this page:
- Published data
- Highlighted treatment failures
- What we’re doing to avoid treatment failures
- The financial case
One of the things that I have always tried to do is to intermittently highlight patients that fail our treatments. Why? Because we don’t do magic pixie dust. We offer medical procedures with success and failure rates just like any other medical procedure. So let’s jump into one of our own Regenexx reviews.
Published Data
If you look at clinics offering stem cell or orthobiologic treatments, while positive testimonials abound, what you almost never see is actual patient-reported data from a registry. In fact, we’re now seeing yet again, calls for a national registry tracking regenerative medicine outcomes. However, we have tracked our patients in a registry going back to 2005.
We have also published that registry data transparently on-line. Meaning, since this is patient-reported information using validated outcome metrics and since we don’t filter it in any way, what you see is what you get. Basically, it’s our medical network publishing its own form of live, online, Regenexx reviews. Let’s take, for example, knee arthritis.
Right now, we have 9,230 Regenexx patients tracked in our patient registry that been treated with the Regenexx-SD bone marrow concentrate procedure (same-day stem cell procedure). If we look further at just the arthritis patients and the Single Assessment Numeric Evaluation (SANE) score, we see:
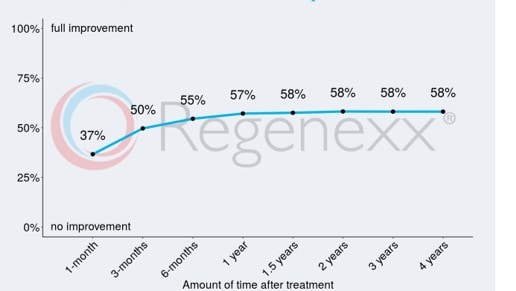
This tells us that the average reported improvement is about 60% better. That means that there are patients in this dataset who had 0% relief of their knee arthritis symptoms. There are also patients who had 100% relief. We could do the same with the other metrics we measure like pain score or the IKDC knee functional questionnaire.
If we look at our published randomized controlled trial on the SD procedure for knee arthritis, what we now call a Perc-TKA procedure, we see that about 7 in 10 patients met the minimum improvement score (MCID) for the KSS questionnaire (1). That means that 3 in 10 didn’t see enough improvement on that functional questionnaire to be certain that they benefitted from the procedure.
Hence, all things being equal, if you have knee arthritis, you have about a 7 in 10 chance of a good outcome and a 3 in 10 chance of the procedure not producing good enough functional results for you to be happy with the procedure.
For other procedures we offer like the Perc-ACLR or Perc-Shoulder Rotator Cuff Repair, the results are better with an even higher number of patients responding. For others, like if you walked in tomorrow with severe hip arthritis, the results are considerably worse. So much so that we try hard to dissuade severe hip arthritis patients from getting a bone marrow concentrate procedure. We’ve published this data as well (2).
Highlighted Treatment Failures
This patient is a 66-year-old man treated by Chris Williams, one of our former fellows who is now in his own practice in Atlanta. First, Chris is one of the best physicians we have ever trained and is so skilled, that I would gladly send my mother to him for treatment. However, whether it’s Chris or myself treating the patient, there are always treatment failures. So let’s dive into one of our own Regenexx reviews on what we offer.
This young elderly man patient had a 5-year history of increasing left than right knee pain with tennis. He first tried a corticosteroid (cortisone) shot, which didn’t help and was taking an NSAID drug when he was first seen. He also tried an umbilical cord “stem cell” treatment which had also failed. The pain was 3-6 out of 10 and his MRI showed partial to full-thickness loss of cartilage with a bone marrow lesion, degenerative medial meniscus tearing, mild swelling, and a Baker’s cyst. His ultrasound exam demonstrated bone spurs and a lax MCL, meaning the knee was also unstable.
The patient was treated with his own bone marrow concentrate using the Regenexx-SD procedure (Perc-TKA) which included injections into the knee joints, ligaments, tendons, and the bone lesion. Initially, he did well, reporting significant relief that allowed him to go back to playing tennis, but after a few months, he backslid and had no relief. PRP booster shots were tried to no avail, so he was eventually declared a treatment failure.
What could have been done differently? I’ve reviewed the case and from Dr. William’s standpoint, nothing. However, from the patient’s standpoint, he refused to participate in the recommended post-procedure physical therapy. Perhaps that would have made a difference, perhaps not, but we do know that active muscular control is one way to help reduce joint instability in addition to ligaments.
What We’re Doing to Avoid Treatment Failures
Many years ago we looked at things like MRIs to see if we could find anything that indicated why one patient was more likely than another to fail the procedure. We also looked at age, sex, weight, etc… (3) However, nothing really correlated with a poor result. Now we’re taking a look again, using different MRI metrics in a research project that should be completed the first quarter of 2020. Will we find something different now? Maybe.
We have also embarked on a multi-year project (now close to completion) where we have examined the levels of 25 cytokines and growth factors in the synovial fluid of the knee and used that information with a machine-learning algorithm to predict failures.
That was promising enough for us to pull the trigger on a bigger study using 440 growth factors and cytokines. That data is now being crunched and if it again looks good, it’s likely we will begin to offer a diagnostic test for patients to undergo before the procedure. That could be ready sometime this next year (2020).
So we are not idly sitting by ignoring the concept of treatment failures. We are actively trying to mitigate their impact by spending heavily to identify who shouldn’t have the procedure. Why? Because that’s the right thing to do.
The Financial Case
Even with treatment failures, we have put together a cost-savings model for employers that shows that we can save more than 40% when the Perc-TKA procedure is used instead of a surgical TKA (knee replacement). See below for how that was calculated:
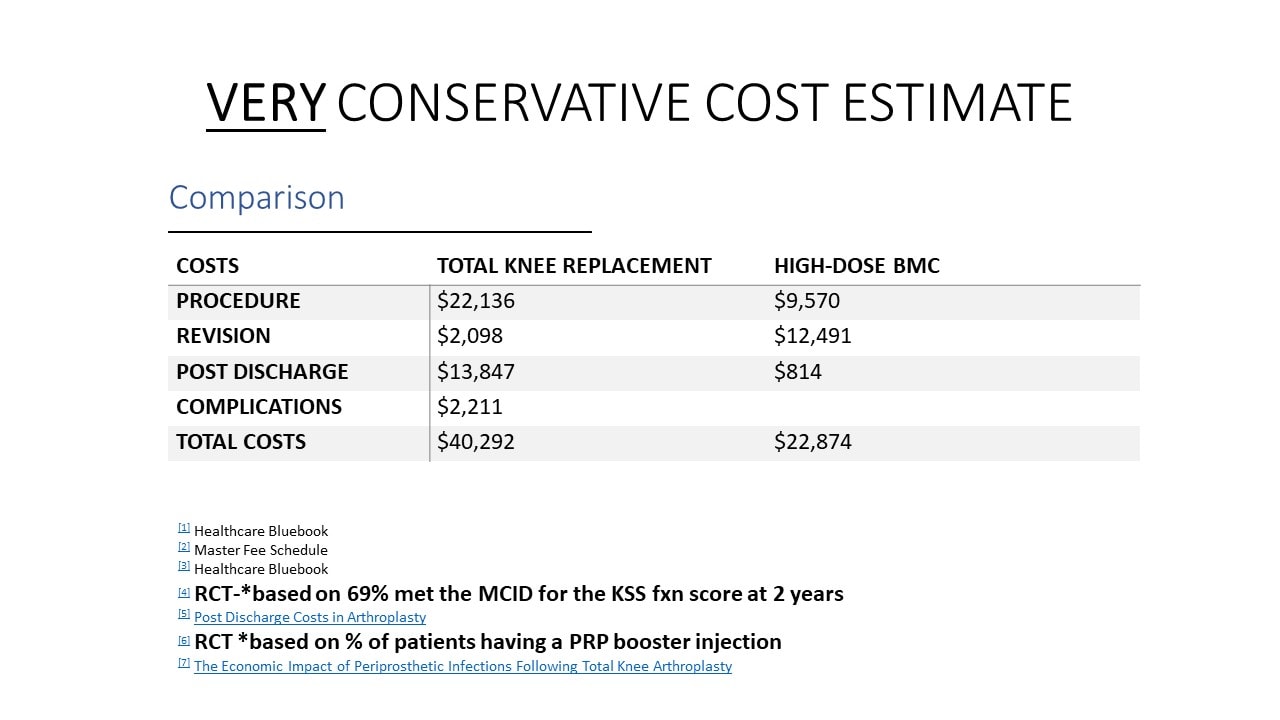
The upshot? What we do isn’t magic. Every procedure we offer, whether it’s platelet based or bone marrow sourced has a success and failure rate. It’s critical that all patients understand that before they decide to undergo a Regenexx or any orthobiologics procedure.
To make sure of that, we publish our results online and in journals and every once in a while I blog on the topic of failures. Basically, a form of Regenexx reviews on what we offer, as, like anything you can buy, all positive reviews usually indicate that the company is gaming the system!
__________________________________
References
(1) Centeno C, Sheinkop M, Dodson E, et al. A specific protocol of autologous bone marrow concentrate and platelet products versus exercise therapy for symptomatic knee osteoarthritis: a randomized controlled trial with 2 year follow-up. J Transl Med. 2018;16(1):355. Published 2018 Dec 13. doi:10.1186/s12967-018-1736-8
(2) Centeno C, Pitts J, Al-Sayegh, Freeman M. Efficacy and Safety of Bone Marrow Concentrate for Osteoarthritis of the Hip; Treatment Registry Results for 196 Patients. J Stem Cell Res Ther. 2014;4:10. https://www.researchgate.net/publication/269277466_Efficacy_and_Safety_of_Bone_Marrow_Concentrate_for_Osteoarthritis_of_the_Hip_Treatment_Registry_Results_for_196_Patients
(3) Centeno C, Pitts J, Al-Sayegh H, Freeman M. Efficacy of Autologous Bone Marrow Concentrate for Knee Osteoarthritis with and without Adipose Graft. BioMed Research International, vol. 2014. doi:10.1155/2014/370621

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
