CoreCyte Update: A First or Second Pinnochio Award?
Ughhh…That’s what just came out of my mouth as I received yet another product packet from the out-of-control birth tissues industry. This time that marketing collateral is on a Wharton’s jelly product called CoreCyte. As a result, I thought I would get started on another CoreCyte review as I’ve looked at this one before. Let me explain.
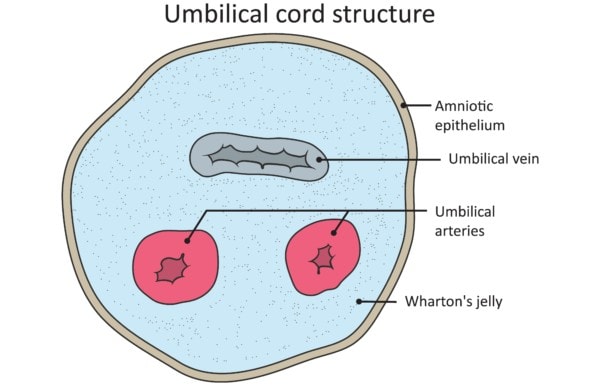
What Is Wharton’s Jelly?
What is Wharton’s jelly? That’s the stuff that keeps the umbilical cord stiff. It’s called that because it’s a jelly-like substance that lives in the umbilical cord and surrounds the arteries and vein located there. In a fresh umbilical cord, there are mesenchymal stem cells in the Wharton’s jelly.
However, it’s very doubtful that any of these stem cells survive in any Wharton’s jelly product on the market today. Why? We’ll get there.
What Is CorCyte?
CorCyte is a commercially available FDA-registered tissue product made from Wharton’s jelly. Since this product is not FDA approved or FDA cleared, but merely registered with a free online form, the agency limits what can be done to the Wharton’s jelly and also requires this to be a dead cell product. To understand more about all of that, see my video below:
CoreCyte’s First Pinnochio Award?
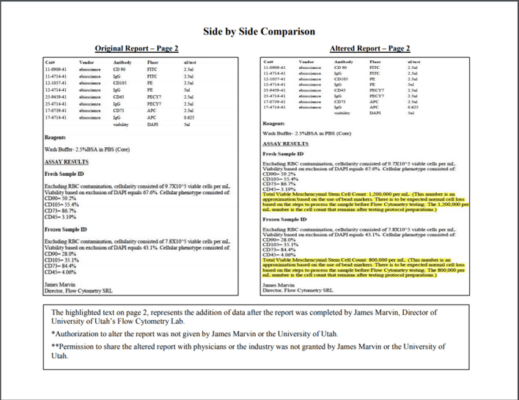
A few years back, I reviewed a flow cytometry report on CoreCyte that was published by the University of Utah. Its conclusions made little scientific sense and they seemed way out there relative to the data published in the report. Then in 2017, I found a document that seemed to be a retraction issued by the University of Utah’s flow cytometry lab. Basically, the document consisted of side-by-side copies of the flow cytometry report on the CoreCyte product, both similar to the one I had previously reviewed. The second copy had a yellow highlighted area that said it had been added and not authorized by the author of the report, a James Marvin, PhD. See below (click on the image to see the PDF):
A sales rep then reached out to me, after I had published the redacted report, saying that the redaction was false. I then called Dr. Marvin at the University of Utah. He confirmed that his retraction was real. Hence, someone had doctored his report.
Understanding Flow Cytometry and This First Pinocchio Award
If you really want to confuse the heck out of a physician, just throw a flow cytometry report his or her way. These simply aren’t tests that 99.9% of physicians ever see on a day-to-day basis, so it’s just noise and a conclusion. Meaning, most doctors won’t have any idea of how to test the accuracy of the report’s data, so they will just read the stuff that summarizes what this all means. So that you can follow along, watch my video below:
Now that you’ve been educated by the video, let’s dive into the the stuff in yellow. What does that say?
Note that in the yellow highlighted area, James Marvin appears to draw conclusions that there are 1.2 million mesenchymal stem cells (MSCs) in a fresh CoreCyte sample and 800,000 in a frozen sample. First, this conclusion makes no common sense based on the data reported as MSCs would have to have CD90, CD105, and CD73 on their surface at the same time. Here these markers are reported separately, but if any conclusion was to be drawn about MSCs, these markers would need to be reported as CD90+/CD105+/CD73+ and then a percentage of cells that had all of these markers.
Second, the simple math makes no sense. Take the fresh CoreCyte sample. They start with 970,000 cells (9.7 x 10 exp 5). We know that MSCs have to have all three of those markers above at once and the least common marker (CD105) represents 55% of the sample. So simply multiplying 970,000 x 0.55=533,500. So how in the heck can you get to 1.2 million MSCs per ml? There weren’t even that many cells per ml of any type in the total sample tested! So even if you had 100% of the cells identified as MSCs, there could only be 970,000 cells per ml. Even the 533K number is way too high, as the cells also need to be negative for other markers.
Third, the viability of both of these samples is awful. 68% from a fresh sample? That number should be >90%, which is what fresh autologous samples run. 43% after freezing and thawing? Again, properly handled frozen stem cell samples should have a viability of >90%.
Fourth, this conclusion is fiction. I called James Marvin and asked him if he concluded that there were MSCs in these samples. I got a resounding no. I also asked him if he authored a retraction. He stated yes. He also relayed that he and the university had gotten threatening legal letters over his retraction.
Finally, I could never confirm through Predictive whether they altered this report. So let’s say that the first Pinnochio award goes to the individual who made these additions in yellow. I also later got threatening legal letters from Predictive over my blog post. So I moved on.
CoreCyte and ISCT
I have mentioned in many blog posts that none of these companies selling amniotic or umbilical cord products meet the guidelines promulgated by the International Society for Cellular Therapy (ISCT) to identify mesenchymal stem cells. This involves not only testing for the presence or absence of specific flow cytometry markers but also for whether these cells are adherent to plastic and form colonies. In addition, more research beyond that must be done to prove trilineage differentiation. I noticed that the ISCT guidelines appeared on the Predictive website shortly after my blogs showing that Predictive and other similar companies didn’t follow these same guidelines. I thought that was odd. To learn more about ISCT guidelines, see my video below (FACT is the accreditation body for ISCT):
The New Predictive Info and a Second Pinnochio Award?
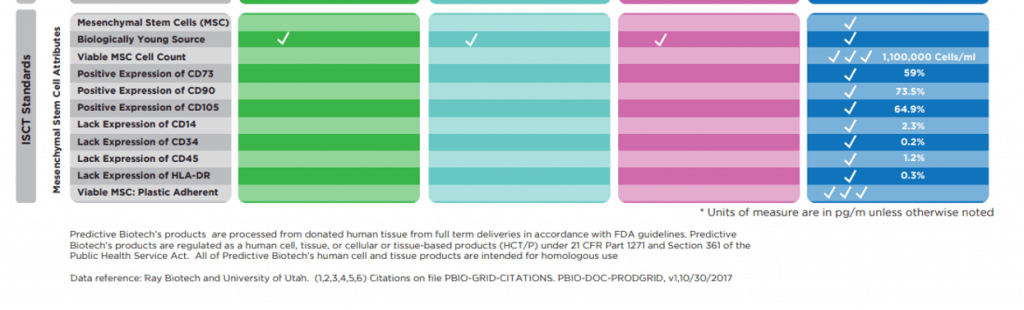
I am sent stuff all the time about amniotic and umbilical cord products. This week, I was sent the new CoreCyte marketing materials and a new product announcement. Turns out Predictive is now marketing a concentrated Wharton’s jelly product. However, more concerning was the marketing flyer sent on CoreCyte. This information is below:
Now we have a whole new set of flow cytometry marker data, but no report to back any of these numbers up. There is also a claim that these cells are plastic adherent and that there are 1.1 million viable MSCs per ml. Huh? That number is curiously close to the 1.2 million cells per ml for the fresh CoreCyte product reported in the bogus University of Utah report. It’s more than the frozen CoreCyte number in that same report, which was 800,000. So how now did we get to 1.1 million viable MSCs? Your guess is as good as mine.
In addition, these flow cytometry markers, as reported, are still not consistent with the ISCT guidelines. In that case, they would need to be reported as CD73+/CD90+/CD105+ (meaning positive for all three markers at once) and then a percentage. We don’t see that here.
In addition, this math is also still messed up. Let’s look at CD34, which is a marker associated with hematopoietic stem cells among other things. The ISCT guidelines say that this should be absent on MSCs and the data here shows that it was absent on 0.2% of these cells tested. That means that only 0.2% of the cells in this sample could be MSCs based on the absence of that marker. Obviously, only some small percentage of those cells would have all of the other markers present or absent. However, let’s assume the best possible (but unrealistic) case for Predictive’s product, which would be that 100% of those CD34- cells were MSCs. Now, we can then back-calculate how many total cells were in the sample. That means that the total cell content of 1 ml as tested would have to be 100/0.2 x 1.1 million. That number is then 500 times 1.1 million MSCs, which is 550 million cells per ml? Having worked with cells for a decade and a half, there is no way to pack that many cells into a 1 ml fluid volume. Let’s say that only 10% of the cells that lacked the CD34 marker were MSCs, then the math gets even more screwed up. Now we have 100/0.02 or 5,000 times 1.1. million.
Maybe there is a dilution factor here at play? Meaning, sometimes cells are tested after dilution. There is no way to tell because there is no supporting documentation.
There is also no information provided in this or any other Predictive document to support that there was any testing performed on CoreCyte on plastic adherence or trilineage differentiation. Curiously, the reference at the bottom says that the supporting citations are stored at “PBIO-DOC-PRODGRID.” What is this? When I Google that or any part of it, I’m brought right back to Predictive Bio’s marketing collateral without any supporting documentation. I also extensively searched the Predictive website, and I also found none of these citations. Finally, I reached out to several people associated with the company to try and get this information. I had not heard back at the time of this writing.
More interesting is that the University of Utah is listed as a reference? When I spoke to James Marvin, he was hopping mad at this company, so it’s hard for me to believe that he did more testing for Predictive. While that’s possible, it seems unlikely.
The upshot? So is this Predictive’s and CoreCyte’s second or first Pinnochio? It’s hard to tell without some supporting documentation. I can say that the University of Utah report circulating around the Internet in 2017 was doctored by somebody. I can also say that the data in this most recent collateral on Predictive’s CoreCyte product doesn’t make sense nor is there any documentation to support that it all adheres to the ISCT guidelines. So we still have a situation that nothing produced by Predictive supports its contention of more than a million live and functional MSCs per vial.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.