My Letter About How SI Joint Fusion Likely Won’t Help Patients in the Long Run
I’ve blogged a few times about why I think the new “minimally invasive” SI joint fusion is not a good treatment option. After that I was asked by a professional organization in Oregon to write a letter explaining my views to their Health Evidence Review Commission, which is considering coverage for the device. This is that letter:
Ariel Smits, MD MPH
Medical Director, Health Evidence Review Commission
RE: “Minimally Invasive Sacroiliac Joint Fusion.”
Dear Dr. Smits
It’s my understanding that your Value-based Benefits Subcommittee is reviewing the data on whether to support reimbursement for minimally invasive sacroiliac joint (SIJ). This is a procedure that I see causing immense, long-term potential harm to patients. Let me explain.
Blamb/Shutterstock
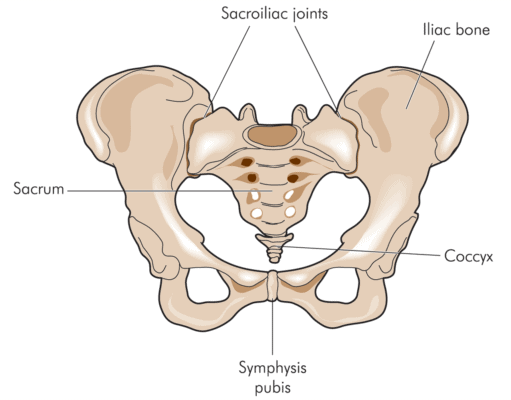
The SI or Sacroiliac Joint
The SI joint resides between the sacrum and ilium—hence the name sacroiliac. The joint has movement, but not like a traditional hinge joint. It’s viscoelastic and “gives” in specific directions and acts as an important shock absorber between the legs and the spine [1]. Basically, it helps transmit energy from the feet to the spine and vice versa.
The joint is ear shaped and is surrounded by strong ligaments that dictate movement. The deep abdominal muscles also assist in stabilizing the joint [1]. The SI joint can be injured by a fall on the buttocks, a car crash, or through other mechanisms [1]. Pain from the SI joint generally localizes on the side of the sacrum at the PSIS. However, symptoms can also mimic radiculopathy, refer into the groin, or to the side of the hip [1, 2]. Physical therapy and chiropractic are common treatments and when those fail, the next most common recommendation is to inject corticosteroids into the joint [3].
Fusing Spinal Joints
I’ve treated patients with post fusion pain and problems my entire career. It’s often personally upsetting to see the walking wounded these procedures produce. As a result of treating hundreds of such surgically disabled patients through the years, I often tell my patients that fusion is a “dog with big fleas.” What do I mean?
The “fleas” are called ASD or adjacent segment disease. This means that the joints above and below the fused joint begin to get painful or break down. This is caused by the fact that energy that should be handled by the fused joint gets shunted above and below to joints that are not prepared to handle those magnified forces.
As an example of how early data on invasive procedures can deceive, spinal fusion for stenosis is important to highlight. Many case series and comparison trials through the years had suggested that lumbar fusion for lumbar stenosis was effective [4]. However, a recent randomized controlled trial showed no efficacy and much higher complications over laminectomy alone [5]. This is consistent with the experience of many clinicians. So while the lower-level research shows promising results, higher-level research shows no efficacy amidst additional patient harm.
For the SI joint, the joint below is the hip, and the joint above is the L5–S1 spinal segment. As discussed, the SI joint is a key energy transfer mechanism, so fusion eliminates that important shock absorber. The hip joint and lower lumbar spine are the next levels that will be overloaded once the SI joint no longer moves.
The Research Supporting Minimally Invasive SI Fusion
Two new studies have now been published which are one and two year follow-ups of patients that were previously enrolled in a randomized controlled trial (RCT) with a 6-month follow-up [6, 7]. Before I review those, let’s examine the results from the first 6-month RCT [8].
The 6-month study was sponsored by the company that makes the extraordinarily expensive hardware to fuse the SI joint. The study seems to show much better results for the SI joint fusion treated group than those patients who were randomized to be treated with physical therapy and SI joint injections. However, the biggest issue with the study is obvious. ASD doesn’t generally show up for several years, so a 6-month RCT doesn’t tell us much.
The randomized controlled trial was abandoned at 6 months as the two new studies only report results for the treated group at 12 months and 24 months (meaning there is no longer a comparison to the patients who only received nonsurgical care; e.g., physical therapy, steroid injections, etc.). So these new studies are not high-level research. I will review the two-year report for ease of discussion.
The two-year study again seems to report good results for patients who had on average about 5 years of SI joint pain before the procedure and who were diagnosed via an image-guided anesthetic block. However, how good is debatable. For example, before the procedure 76% of the patients needed to take narcotics for SI joint pain, and two years after the procedure, 55% were still taking narcotics. Given the significant morbidity caused by long-term narcotic use, reducing that use by merely 21% with more than half of the treated patients still opioid dependent is a significant problem.
The study reports about 5% of the patients had serious side effects, but is that number accurate? Another new study, not sponsored by the manufacturer, refutes that data [9]. The new study was not paid for by the company. In fact, it’s a search of an insurance company database of the claims and complications paid by Humana on more than four hundred SI joint fusion procedures. At 6 months, the complication rate in this study was more than three times higher than that reported in the device company sponsored study (4.7% vs. 16.4%) [9]! The insurance study reports that a significant number of patients are reporting new spinal problems (i.e., the fleas) after the surgery.
Financial Bias
A search of the nonprofit ProPublica website (see https://projects.propublica.org/docdollars/) shows that the lead author of the new two-year study was paid $72,007 by IFuse Implant, the maker of the SI fusion device under review before the state of Oregon. The second author was paid $34,296 by the company. Interestingly, the full text copy of the two-year paper doesn’t disclose if either of these lead authors owns any stock, stock options, or other equity in the company.
Why this is likely an Unnecessary Surgery
When all you have is a hammer, everything looks like a nail. Meaning that surgeons tend to try to find surgical solutions for problems rather than nonsurgical. Chronic SI joint pain is no different. Let me explain.
Prolotherapy (prolo) is a simple regenerative-medicine technique that’s been used since the 1940s to treat SI joint pain. Because it has no business model (i.e., a hyper-expensive drug or device that can garner huge insurance reimbursement), there has never been the funds needed to get this simple treatment approved by insurance companies.
Prolotherapy treatment for the SI joint would involve injecting the loose ligaments that are causing instability with the goal being to tighten those, rather than fusing the joint. The interesting thing here is that there already is a randomized controlled trial comparing prolotherapy to a steroid injection of the SI joint showing similar and/or superior results to the studies paid for by the SI joint fusion device company [10]. While it involved fewer patients, it followed patients for much longer in a controlled trial (15 months vs. 6 months) than the SI joint fusion device. The prolotherapy study demonstrated stark superiority over the steroid injection. The cost? A few hundred dollars an injection, a small fraction of the money that will be paid by the State of Oregon for the expensive SI joint fusion implant, surgeon’s fees, surgery center or hospital fees, anesthesia, and complications generated by same.
In conclusion, by the state of Oregon approving compensation for this procedure, I fear that thousands of Oregonians will be needlessly harmed. In addition, cheap therapies that have the same evidence base as this procedure and have none of the side effects of fusing the SI joint are already widely available; hence, there is no need to approve compensation for this invasive and expensive procedure.
Christopher J. Centeno, M.D.
References:
- Richard L. DonTigny, B., PT, Dysfunction of the Sacroiliac Joint and Its Treatment. The Journal of Orthopaedic and Sports Physical Therapy, 1979. 1(1): p. 13.
- Slipman, C.W., et al., Sacroiliac joint pain referral zones. Arch Phys Med Rehabil, 2000. 81(3): p. 334-8.
- Laslett, M., Evidence-based diagnosis and treatment of the painful sacroiliac joint. J Man Manip Ther, 2008. 16(3): p. 142-52.
- Weinstein, J.N., et al., Surgical versus nonoperative treatment for lumbar spinal stenosis four-year results of the Spine Patient Outcomes Research Trial. Spine (Phila Pa 1976), 2010. 35(14): p. 1329-38.
- Forsth, P., et al., A Randomized, Controlled Trial of Fusion Surgery for Lumbar Spinal Stenosis. N Engl J Med, 2016. 374(15): p. 1413-23.
- Duhon, B.S., et al., Triangular Titanium Implants for Minimally Invasive Sacroiliac Joint Fusion: A Prospective Study. Global Spine J, 2016. 6(3): p. 257-69.
- Duhon, B.S., et al., Triangular Titanium Implants for Minimally Invasive Sacroiliac Joint Fusion: 2-Year Follow-Up from a Prospective Multicenter Trial. Int J Spine Surg, 2016. 10: p. 13.
- Whang, P., et al., Sacroiliac Joint Fusion Using Triangular Titanium Implants vs. Non-Surgical Management: Six-Month Outcomes from a Prospective Randomized Controlled Trial. Int J Spine Surg, 2015. 9: p. 6.
- Schoell, K., et al., Postoperative complications in patients undergoing minimally invasive sacroiliac fusion. Spine J, 2016.
- Kim, W.M., et al., A randomized controlled trial of intra-articular prolotherapy versus steroid injection for sacroiliac joint pain. J Altern Complement Med, 2010. 16(12): p. 1285-90.”

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
