Exploring Fibrin Disc Injection For Degenerated Discs

Medically Reviewed By:
Degenerated discs often cause persistent symptoms that interfere with daily activities, work responsibilities, and overall quality of life. Traditional treatments, such as medications or surgery, do not always promote lasting improvements in function or provide consistent symptom management.
Fibrin disc injection provides a less invasive option compared to conventional surgical treatments. This treatment supports the structure of the affected disc and promotes improved function by addressing contributing factors related to disc degeneration rather than simply masking symptoms.
What Is Disc Sealing With Fibrin?
Some physicians perform disc-sealing procedures by injecting fibrin, an insoluble protein the body naturally uses for blood clotting and tissue repair. In these treatments, concentrated fibrin acts like a biological adhesive, functioning similarly to rubber cement. Physicians inject it into the disc or its tough outer layer (annulus fibrosus) to seal small tears or holes.
The procedure aims to reduce the risk of internal disc material leaking out by sealing the disc. Supporters of this approach suggest that when internal disc materials leak, they may expose nearby spinal nerves to chemical substances, causing inflammation. This inflammation process, called chemical radiculitis, has been associated with symptoms in the back or legs.
Some physicians may also use fibrin sealing before performing procedures that use bone marrow concentrate (BMC) or platelet rich plasma (PRP). Sealing the disc first may help retain the injected material within the disc structure, to support better integration and promote improved outcomes.
How Herniated Discs Affect The Spine
Discs serve as shock absorbers between the bones of the spine. Each disc contains a soft inner core, called the nucleus pulposus, surrounded by a tough, fibrous outer layer known as the annulus fibrosus. The structure of discs allows the spine to bend and move while protecting it from stress.
Over time, discs can lose water content and become less flexible. This drying out, called dessication, weakens the disc and makes it more prone to damage.
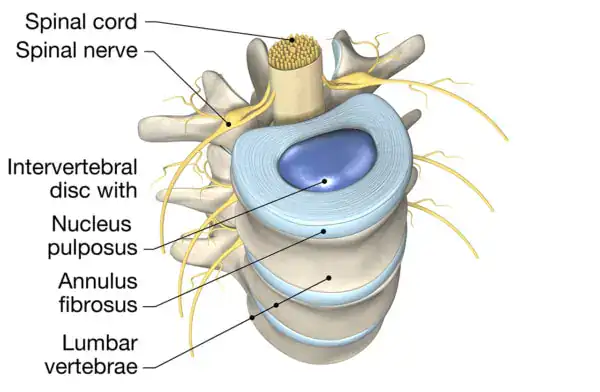
With repeated stress and declining support, small tears may develop in the outer fibrous layer.
When these tears form, the soft inner material may push through weakened areas of the annulus fibrosus. This process, known as herniation, can place pressure on nearby nerves and contribute to symptoms such as pain, numbness, or weakness in the affected areas.
When Is Fibrin Injection Recommended?
Physicians may consider fibrin disc injection when small tears or defects in the outer layer of a spinal disc are suspected of contributing to ongoing symptoms. These symptoms may include localized back pain or radiating discomfort linked to irritation of nearby spinal nerves.
This approach is usually considered when conservative treatments—like physical therapy, medications, or exercise—haven’t provided enough relief. In these cases, sealing the disc with fibrin may help stabilize the disc and reduce irritation caused by chemical inflammation.
Physicians may also use fibrin sealing in preparation for other interventional procedures. For example, when planning to place BMC or PRP into a disc, sealing the disc first may help retain the injected material and support the treatment process.
Not every case of back pain or disc injury is appropriate for fibrin injection. Careful diagnostic evaluation, including imaging studies and a clinical assessment, remains essential before proceeding with this approach.
How Is Fibrin Injection Administered?
Physicians typically perform fibrin injections using precise image guidance, such as fluoroscopy, to accurately place the material into the targeted area of the disc. The physician carefully advances a small needle into the disc space or any identified tears within the outer layer.
Once positioned, the physician injects contrast followed by a fibrin sealant. The material flows into small cracks or holes, forming a soft, flexible barrier. This sealing process aims to reduce disc material leakage and support the disc structure’s overall integrity.
Following the procedure, a gradual reduction in symptoms may be experienced as the disc stabilizes and chemical irritation decreases. Some individuals may notice reduced back pain or relief from nerve-related symptoms over time; however, responses can vary based on the extent of disc damage and the presence of other spinal conditions.
Fibrin injection may complement broader treatment strategies rather than serve as a stand-alone solution. Post-procedure rehabilitation and ongoing monitoring often play essential roles in supporting recovery and promoting improved functional outcomes.
Possible Risks And Side Effects Of Injectable Fibrin
Like any procedure involving biological materials, fibrin injection may carry potential short-term and long-term risks. While serious complications appear uncommon, individuals and physicians should carefully consider important factors when evaluating this approach.
Using donor-derived biologic materials may involve a small risk of disease transmission, even with appropriate screening. Physicians and individuals should weigh these considerations alongside the potential benefits of treatment.
Potential short-term and long-term effects of injectable fibrin may include:
Effects Of Fibrin On Disc Height
Before-and-after imaging following injections into a disc often shows an initial increase in disc height. Physicians have observed this effect with fibrin injections, same-day BMC procedures, and PRP treatments. The disc may appear to “plump up” due to the presence of increased fluid pressure after injection.
However, this increase in disc height may be temporary, particularly in severely degenerated discs. As the body dismantles the newly formed fibrin, usually within one to two weeks, the disc may return to its prior collapsed state. Although early imaging may show improved height after injection, these changes do not usually persist over time and should be interpreted with caution.
Effects On Disc Integrity
Fibrin injection may temporarily support the structure of a damaged disc by sealing small tears and reinforcing weakened areas. This mechanical support can help stabilize the disc in the short term, potentially reducing chemical irritation and nerve-related symptoms immediately following the procedure.
However, discs affected by significant degeneration often present deeper structural challenges beyond surface tears. Over time, as the injected fibrin material breaks down, the disc’s original weaknesses may reappear. Without addressing the broader mechanical instability, the disc may continue to collapse or degenerate despite initial improvements.
Additionally, there’s research supporting a concern that the introduction of a needle into the disc may increase the risk of disc degeneration.
Infection
As with any procedure that penetrates the body’s natural barriers, fibrin injection carries a potential risk of infection. Although infections following disc injections remain rare, they can occur if bacteria enter the disc space during the procedure or if the disc environment becomes compromised.
An infection within the disc, known as discitis, may cause significant symptoms, including worsening back pain, fever, and difficulty moving. In severe cases, discitis can lead to spinal instability or require additional medical intervention, such as prolonged intravenous antibiotic therapy or surgery.
Disc Reherniation Reported In Some Cases
In some cases, physicians have reported disc reherniation following fibrin injection. While the initial sealing effect may help stabilize small tears, it does not always prevent disc material from reemerging through weakened areas over time.
Several factors may contribute to reherniation, including the degree of underlying disc degeneration, mechanical instability within the spinal segment, or incomplete healing of the disc structure after the injection. Physical stress on the spine after treatment may also disrupt the sealed area and increase the risk of recurrence.
Links To Chronic Condition Risk In Some Cases
In rare situations, complications from fibrin injection may contribute to the development of chronic spinal conditions. Chronic discitis, progressive disc degeneration, or long-term nerve irritation may develop if the disc does not heal properly after the procedure. The risk of chronic complications tends to be higher in individuals who have significant pre-existing disc damage or spinal instability. If infection, persistent inflammation, or repeated reherniation occurs, these factors can place ongoing stress on the spine and surrounding tissues.
Misconceptions Between Disc Sealing And Regenerative Medicine
Several misconceptions have emerged regarding fibrin disc sealing and regenerative medicine procedures. Promotional materials for the Pauza Discseel™ procedure claim support from a randomized controlled trial. However, the only trial conducted found no greater benefit than a placebo injection.
Promotional sources have also claimed that sealing a disc before a BMC injection is necessary to prevent cancer caused by leaking stem cells. No credible scientific evidence supports this claim. A safety study of 2,372 patients, including individuals who received BMC injections into discs, reported no cases of cancer development.
Another misconception on why disc sealing is needed prior to BMC injection includes a risk for developing bone spurs. Unfortunately, this is based on a study that does not accurately replicate the consequences of a torn disc in humans (cite).
Assertions based on such studies fail to accurately represent the nature, clinical safety, and application of modern procedures using Regenexx lab processes.
Is There An Alternative Approach To Managing Degenerated Discs?
Procedures using Regenexx lab processes offer a non-surgical alternative to disc sealing with fibrin by using a patient’s own bone marrow concentrate (BMC) to support disc structure and function. This approach minimizes reliance on donor-derived materials and the potential risks associated with them.
Instead of focusing on mechanically sealing the disc, these procedures aim to address the underlying biological factors that contribute to degeneration—such as cell loss, dehydration, and tissue breakdown. By supporting the body’s natural repair processes, physicians in the licensed Regenexx network use image-guided techniques that may help reduce pain and improve function without removing or sealing disc tissue
Other Conditions That May Be Addressed With The Regenexx Approach
While managing degenerated discs remains a primary focus, physicians within the licensed Regenexx network also apply these procedures to a range of musculoskeletal conditions. These conditions often involve injuries or degenerative changes, where supporting the body’s natural healing processes may help improve function and reduce symptoms.
Case Study: Ted’s Experience With Non-Surgical Recovery
Ted’s case illustrates how procedures using a patient’s own healing agents may support disc recovery without requiring additional sealing with fibrin. Ted experienced chronic low back pain for several years and did not improve with physical therapy or epidural steroid injections. His symptoms significantly limited his ability to perform daily activities, including playing with his children.
An MRI revealed a tear in the back of the L5–S1 disc. A discogram confirmed that the L5–S1 disc was painful and leaking, as dye injected during the test immediately escaped the disc space. Although the L4–L5 disc also showed leakage, it did not reproduce symptoms during testing.
In May 2015, Ted underwent a same-day BMC procedure using Regenexx lab processes, targeting the L5–S1 disc with his own BMC. He reported a 75–80% improvement following the initial procedure. Seeking further recovery, Ted received a second injection into the L5–S1 disc in November 2015.
At his one-year follow-up, Ted reported a 95% overall improvement. He had returned to full activity without significant pain, even though fibrin sealing was not used during his treatment.
This case demonstrates how same-day BMC procedures may naturally create a fibrin scaffold when exposed collagen fibers are present within a damaged disc. Unlike dense fibrin glues, which may form a less supportive environment for cellular activity, a patient’s own bone marrow concentrate provides a biological matrix that may help support the body’s natural healing processes.
Individual outcomes may vary. For more information about patient outcomes, please visit our Patients’ Results page: https://regenexx.com/results/.
Make An Informed Decision About Your Disc Care
Current evidence does not support a pressing need to seal a disc with fibrin before performing regenerative procedures. Claims suggesting that sealing prevents serious complications are not substantiated by credible research.
When considering treatment options for disc injuries, using a patient’s own cells remains an important principle. Patient-centered, research-based approaches, such as those utilized by physicians within the licensed Regenexx network, are designed to work with the body’s natural healing processes and may help promote improved long-term function.

Medically Reviewed By:
