A2M Knee Injections: What’s in Your Arthritic Knee?
Regenexx is unique in the world as when everyone else is just beginning to use stem cells to see if they may help, we’ve been refining the science behind this technology for a decade. One of the fascinating things we’ve been doing lately is taking synovial fluid samples from patients before and after our procedures and looking at 25+ cytokines and growth factors. One of those really interesting molecules is called A2M. Let me explain.
First, we call our most recent massive research study our knee micro environment (ME) study (this is the second phase of this research). If you think about how modern farmers measure what’s in their soil before planting their seeds, this is why we’re looking at the cytokines and growth factors in the knee before and after our procedure. We want to learn what needs to be in the “soil” to ensure a great outcome for the patient.
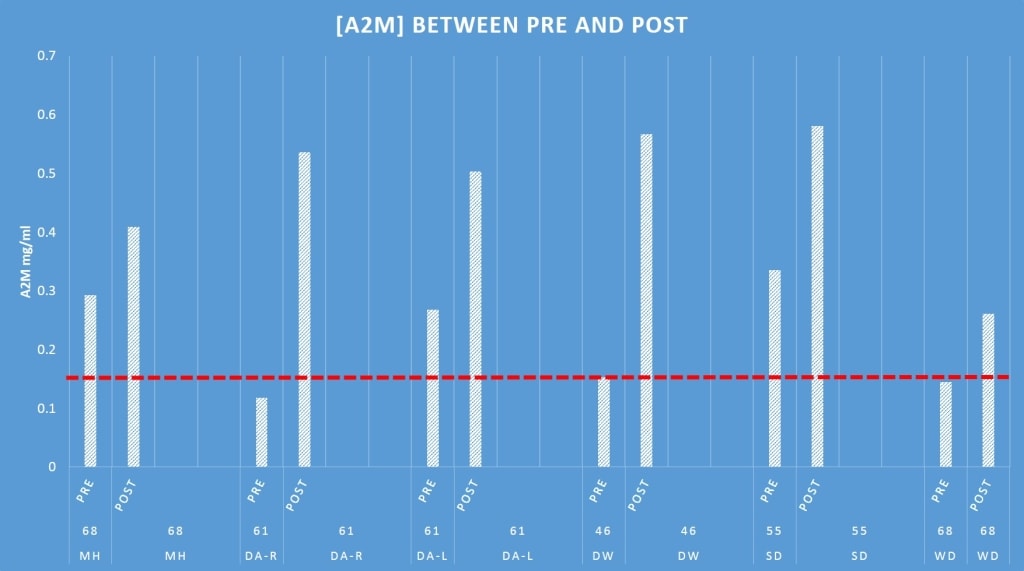
Second, A2M or alpha 2 macroglobulin is an interesting molecule involved in knee arthritis. It’s one of several cytokines in joints that can block the breakdown of cartilage. The others are called IRAP and TIMP, one of which we’re also measuring. A recent trend in regenerative medicine is using bedside machines that can concentrate A2M in serum to help reduce arthritis symptoms through A2M knee injections. While we have no idea of the amount of A2M needed to inhibit arthritis in real patients or even if that happens, we do know from an animal study that levels of about 150 mcg/ml can accomplish that feat in rabbits.
Above is a graph of the A2M levels found in the knee before and after a Regenexx-SD procedure in several patients (n=6). What’s interesting is that about half of the patients exceed the minimum level of A2M in their joint before the procedure and about half don’t have that much or are right at the line (the red dashed line is about 150 mcg/ml). What’s also interesting is that 2-4 days after the stem cell procedure, the A2M levels in all patients far exceeds the minimum amount. We believe this is a good thing and that this is likely explained by the fact that the proprietary type of bone marrow concentrate we’re using has high levels of A2M, so we’re just measuring what was placed in the joint a few day’s earlier.
There are still a bunch of unknowns here. Does the level of A2M matter at all when it comes to the final outcome of the treatment? Does not having enough at the start portend a bad or good outcome? While we know how much you need in a rabbit, do you need more or less in a human? Our knee micro environment study should help answer these questions.
The upshot? At Regenexx we’ve been advancing the science behind real world orthopedic stem cell treatments for a decade. The fact that our knee ME study is breaking new ground in our understanding of how best to improve the clinical results for Regenexx procedure patients is game changing. Over the next 6 months we should begin to finally figure out how best to fertilize the soil and continue to eliminate the guesswork so prevalent in stem cell treatments.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.