Is Injecting the Bone in Knee Arthritis the New Standard?
If you read this blog, you know that there are VAST differences in knowledge and training between providers offering knee “stem cell” treatments. In addition, what you get at one clinic will be vastly different, and many times not even be similar to what you get at another. The one equalizer is supposed to be the high-level research being published. Hence this morning we’ll talk about two brand new studies that show that bone stem cell injections provide longer relief for knee arthritis patients.
The Knee Arthritis Stem Cell Wild West
If you sign up for a partial meniscectomy procedure, you’re likely to get 90% the same procedure no matter which orthopedic surgeon you see. Hence, while there may be small differences in technique or the tools used, you can rest assured that the procedure is standard. However, walk into 10 different clinics offering “stem cell therapy” for your knee and you’re likely looking at least a half dozen very different procedures all being called the same thing. Meaning that outside of networks like Regenexx, there is ZERO standardization. Let’s dive into this concept.
Here are the main types of procedures out there that fall into the category of knee stem cell procedures:
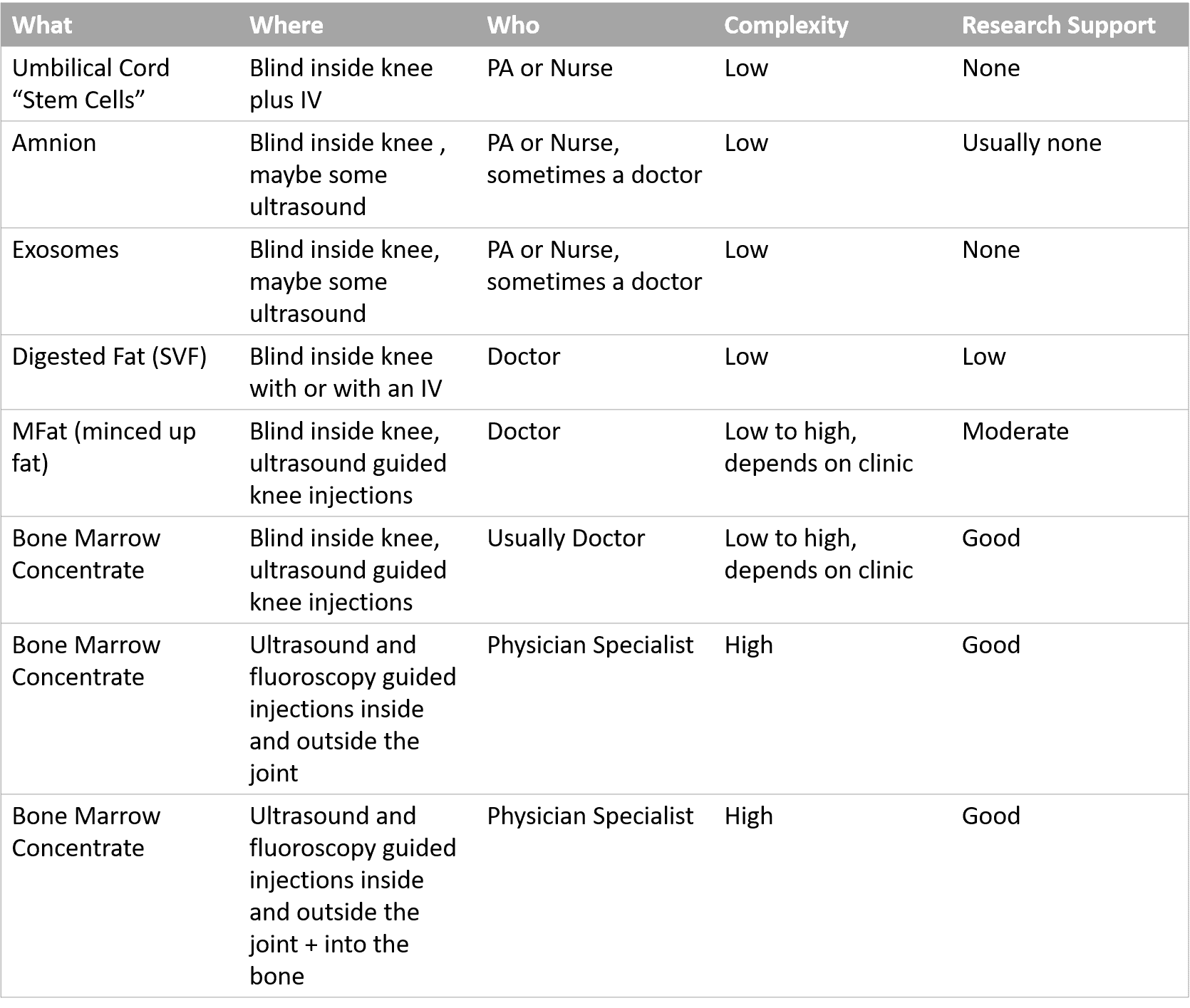
As you can see, these are vastly different procedures that have little in common that are all generally called the same thing. In addition, for most, there is no or little research support that they work. Finally, nothing about their prices line up with the quality or complexity of what’s being done, instead, the price is only related to what the traffic will bear.
RCTs on Knee Arthritis and Stem Cells
The gold standard in medical research is called a randomized controlled trial. That means that the patients are randomly treated with different things and then their results are measured. Sometimes a placebo is used and other times more traditional treatments are compared against new ones.
Up until recently, the only technology to date that had a reasonably powered randomized controlled trial that demonstrated long-term positive results in treating knee arthritis was injecting your own bone marrow concentrate into the knee joint (1). That was our study performed using the standardized Regenexx technique. However, that changed with two new studies focused more on injecting the bone than the joint. Let’s dig in.
The author of both studies is Phillipe Hernigou in Paris, France. Phillipe has been using bone marrow concentrate (BMC or the stem cell fraction isolated from your own bone marrow) since the late 1980s. His focus has been mostly on injecting the bone itself, so he has published seminal papers on things like using BMC to treat avascular necrosis of the hip. Now he has extended that same concept to patients with knee arthritis.
Study 1-Bone Only versus Intra-articular Only Injections
Between the years 2000 and 2005, 60 patients with bilateral moderate knee arthritis were treated. Each had one side randomly treated with a simple injection of BMC inside the knee joint (intra-articular or IA) while the other knee had an injection of BMC only inside the bone (intraosseous or IO) (2). Over 15 years, about 5% per year of the IA group converted to a knee replacement whereas only about 1% of the IO group needed a knee replacement. By the end of the 15 years, this amounted to 70% of the IA group needing a knee replacement whereas only 20% of the IO group did.
This fits with our experience as well, in that the average duration of an IA BMC injection is 2-7 years, which includes bone on bone patients (more severe than were treated in the above study). What’s surprising here is that aggressive treatment of the bone alone worked so well for more than a decade.
Study 2-Bone Only versus Knee Replacement
This study had 140 patients who were elderly (aged 65 to 90 years) (3). These were patients who had been deemed knee replacement candidates for bilateral medial knee arthritis (inside knee). The patient was randomized to have either one side injected IO with BMC or get a knee replacement so that one side compared an injection to the other side which had surgery. At an average of 15 years later (range 10-20 years), only 18% of the IO knees had converted to a knee replacement on that side. Surprisingly, the number of knee replacement knees that needed second knee replacement surgeries (revisions) was almost as high as the number of IO knees that converted to knee replacement.
Regenexx and IO Therapy
Regenexx has been performing IO therapy in selected knee arthritis patients for years and has an upcoming accepted paper on the topic that because of the pandemic, likely won’t get published until early next year. However, based on the long-term results from these two new studies, we have decided to offer all patients who are knee replacement candidates the addition of IO BMC treatment.
Is this procedure painful? We use micro trocars to inject the bone under fluoroscopy guidance. Interestingly, placing those isn’t something the patient often feels when the skin and soft-tissues are numbed. However, injecting the bone marrow can be uncomfortable, so in Colorado, we offer patients IV sedation where they often don’t recall the procedure. Please enquire with each Regenexx clinic as to what they will use for anesthesia to keep you comfortable.
Buyer Beware
One of the things every consumer with knee arthritis should be cautious about is that the stem cell content of the IO injections in these studies was very high. Meaning that most clinics that use quickie bone marrow aspirations and simple bedside centrifuges likely can’t easily achieve these stem cell concentrations. That isn’t an issue at Regenexx clinics as we have been specialists at performing high yield bone marrow aspirations and high concentration bone marrow for a decade and a half.
The upshot? These two new RCTs are game-changers for those of us who use bone marrow concentrate to help our patients avoid knee replacement. As you can see above, it’s the wild west out there and what you get when you walk into a clinic may not be even in the same neighborhood as what’s described in these studies. Hence, buyer beware!
________________________________
References:
(1) Centeno C, Sheinkop M, Dodson E, Stemper I, Williams C, Hyzy M, Ichim T, Freeman M. A specific protocol of autologous bone marrow concentrate and platelet products versus exercise therapy for symptomatic knee osteoarthritis: a randomized controlled trial with 2 year follow-up. J Transl Med. 2018 Dec 13;16(1):355. doi: 10.1186/s12967-018-1736-8. PMID: 30545387; PMCID: PMC6293635.
(2) Hernigou P, Bouthors C, Bastard C, Flouzat Lachaniette CH, Rouard H, Dubory A. Subchondral bone or intra-articular injection of bone marrow concentrate mesenchymal stem cells in bilateral knee osteoarthritis: what better postpone knee arthroplasty at fifteen years? A randomized study. Int Orthop. 2020 Jul 2. doi: 10.1007/s00264-020-04687-7. Epub ahead of print. PMID: 32617651.
(3) Hernigou P, Delambre J, Quiennec S, Poignard A. Human bone marrow mesenchymal stem cell injection in subchondral lesions of knee osteoarthritis: a prospective randomized study versus contralateral arthroplasty at a mean fifteen year follow-up. Int Orthop. 2020 Apr 23. doi: 10.1007/s00264-020-04571-4. Epub ahead of print. PMID: 32322943.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
