Making Meniscus Tissue Better with Stem Cells
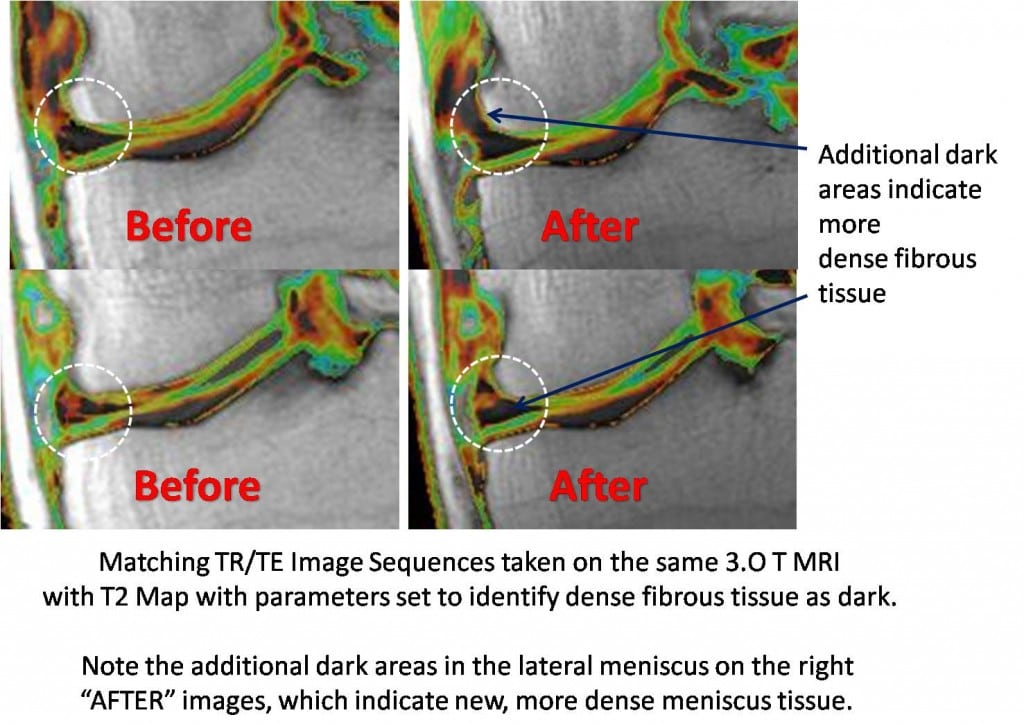
The function of the meniscus is to act as a shock absorber and protect the cartilage. Imaging the meniscus on MRI is difficult. On traditional MRI images it generally shows up as a dark triangle shape. However, accessing it’s composition on traditional MRI images is difficult at best. As a result, we have moved to using ultra high field MRI T2 maps to better characterize the quality of the meniscus tissue. This method has been used in the research for years to access the quality of knee cartilage. This MRI is a case in point. This T2 map is on SK’s lateral meniscus (SK was the subject of yesterday’s post). The parameters of the post-processing have been changed to show dense fibrous tissue (such as ligament and good quality meniscus tissue) as dark. You can see above that before the stem cells are used, his lateral meniscus (broken dark and green triangle in the two “BEFORE” slices above, surrounded by the dashed white circle) has less dark area (good quality dense meniscus tissue) than the “AFTER” images above (which have more dark areas). This may be another explanation why his knee is 100% better after the stem cell re-implant. We have seen other MRI changes in meniscus tissue after stem cell knee injections. Many man hours have been spent trying to figure out how to surgically transplant donor or artificial meniscus tissue into the knee, as the restoration of this tissue as a shock absorber could do wonders to reduce knee pain and protect remaining cartilage. Could it be as easy as a few Regenexx-C procedure knee injections? Not sure, but this data is encouraging.
This patient was treated with the Regenexx-C (cultured stem cell injections)

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
