The Impact on Orthopedic Stem Cell Research of the Slow Death of Embryonic Stem Cell Research
When the Bush white house shut down embryonic stem cell (ESC) research in the early 2000s, the field of adult stem cell research was in it’s infancy. California prop 71 was born, promising embryonic stem cell research breakthroughs that would quickly have the blind seeing and the crippled walking. This never happened, instead, as discussed elsewhere, the resulting California Institute for Regenerative Medicine (CIRM) became a way to keep ESC grant dollars flowing for basic research with little incentive to use those monies to cure disease or treat patients. In the meantime a funny thing happened, adult stem cell research through mesenchymal stem cells (MSCs) was launched. MSCs have now surpassed ESCs as the dominant form of stem cell research. This has big implications for orthopedic stem cell research, as the English say, the King is dead, long live the King.
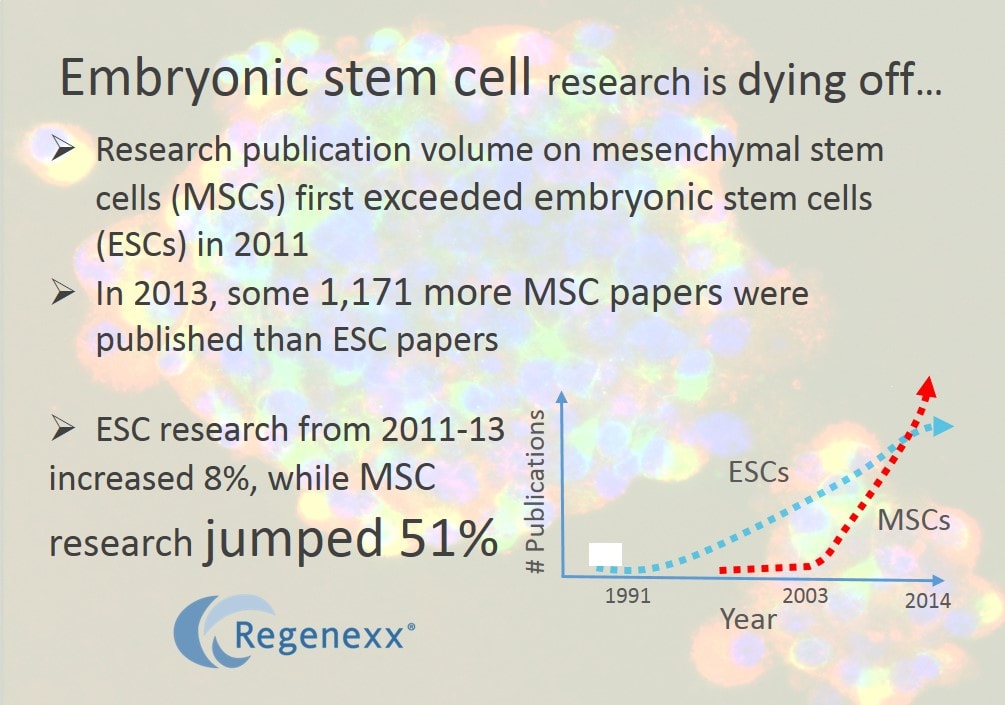
Embryonic stem cells are the cells that create a new fetus. They can be harvested from embryos and ESCs have the advantage of being able to differentiate into most any cell type in the body. However, the fact that human embryos need to be destroyed to create these cells has caused ethical concerns. Because of the government ban on new stem cells lines, in the early 2000s a much less prominent area of research using adults stem cells (MSCs) was catapulted forward. These are cells taken from adults and so they have none of the ethical concerns that limited ESC research. For the last 5-7 years the amount of research being published year after year on MSCs has outpaced ESCs, but in 2011 the adult stem cell research community hit a milestone. In that year more research was published on MSCs than ESCs. In addition, from 2011-2013, while the pace of ESC research has leveled off and is basically flat, the pace of MSC research continues to explode (an 8% increase in ESC research vs. a 51% increase in MSC research). Finally, sometime in the next 12-24 months, the total number of publications on ESCs will be eclipsed by the total number on MSCs (today they stand at 33,404 for ESCs and 29,234 for MSCs). This is quite a feat, given that ESC research first took off back in the early 90s with about a 10 year head start of the inflection point for MSC research (which happened in the early 2000s).
The implications for orthopedic stem cell research of this changing of the old guard for the new is huge. ESC research tended to be more focused on neurologic applications, while MSC research has always had a bigger orthopedic focus. For example, while as of today there are 430 papers on ESCs and cartilage, there are 2,684 for MSCs and cartilage. So the refocusing of resources away from ESCs toward MSCs will have big positive impacts on orthopedic stem cell research.
The upshot? Should we be saddened that ESC research is being eclipsed? Not so much. MSCs and other adult stem cell types can also do amazing things. However, the story of how ESC research was hyped and then never delivered and got knocked off it’s throne is in itself a cautionary tale. Sometimes science proceeds to benefit the interests of science more so than society. In the future, we need to be careful that science serves society and not the other way around!

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.