Regenexx Is Awarded Another Patent
I just came back from Cayman, and yesterday was my first day back in the Colorado office. Waiting for me there on my desk was a bright, shiny, and new patent from the US government. Even though this is one of many we have received, I thought a close-up of the seal on the document would be cool to share.
Regenexx Is VERY Different
In today’s world of the stem cell wild west, where everyone and his brother is offering some faux stem cell therapy, Regenexx has always been and will always be VERY different. As I showed you a few days ago, it’s very different in how the cells are taken and precisely placed back into the body. In addition, it’s very different in how we process the cells using our same-day lab technology.
Bone marrow concentrate is what’s used in a same-day bone marrow-based stem cell procedure. This product has been created the same way since the ’90s. You centrifuge the bone marrow aspirate, which creates a number of layers. You then take out the middle layer, known as the buffy coat, which is where the stem cells concentrate.
Simple Bedside Machines
Almost all physicians who perform bone marrow-based procedures use a simple bedside system to produce bone marrow concentrate (BMC). These are very convenient, as all the doctor or his or her assistant has to know is where to place the bone marrow aspirate (BMA) and the location of the “On” button. The downside is that each machine has to simplify what a technician does manually. This means that corners are cut. For example, the machine often has to include much more of the layers above and below the buffy coat as the size and location of this varies with patient hydration and blood density (hematocrit). Hence, what the machine produces is a much less concentrated and much less pure version of the buffy coat.
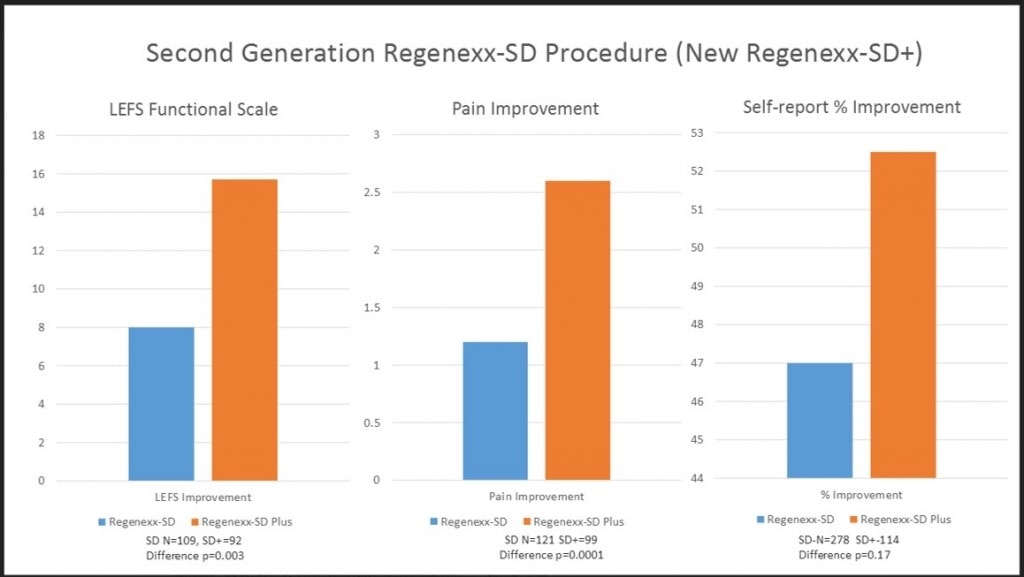
Improving On an Old Technique
We began working on an improvement for the bone marrow concentrate protocol many years ago, in 2011. We discovered techniques to isolate new sources of mesenchymal stem cells in bone marrow and soon submitted a patent, the one that was officially granted this summer. This technique also produced better clinical results when we compared our registry data from before the new procedure to after the higher-yield bone marrow processing was in place (see below).
Over the past several years, we have been granted a bevy of patents on various parts of our three-step process. This includes a patent on our inflammatory preinjection used to create a favorable environment for stem cell-mediated repair and the use of nanogram corticosteroid to push cells toward chondrogenesis. Hence, all parts of the Regenexx process are now protected by US patents.
The upshot? Regenexx is very different. From our lab-based and clinical research program to our clinical registry that tracks outcomes and complications to our patents, we invented this space and continue to record milestones, like insurance coverage. I want to thank our science team and Neven Steinmetz for her dedication in seeing these patents through the process to get many of them granted.

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
