Update on Stem Cells for Severe Ankle Arthritis
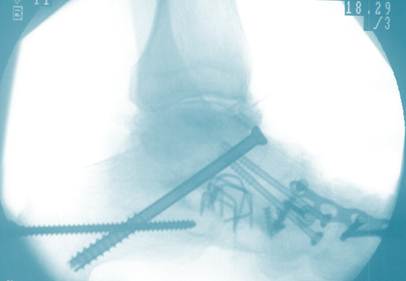
One of the things we pride ourselves on is that we discuss all of our stem cell outcomes and publish our work, which includes the patients that do great with stem cells and the patients that fail the therapy. To that end, I also blog on patients where the procedure either didn’t work or perhaps didn’t result in a permanent fix. HK is an elderly man who was featured in the blog because of the severity of his ankle arthritis and our amazement that the Regenexx-C stem cell treatment restored ankle function and dramatically reduced his pain. His x-ray is above and it doesn’t take a trained medical professional to note that he has a severe ankle problem as indicated by the amazing “hardware store” that was placed into his ankle before we first evaluated him. He was treated in Spring of 2009 and his pain relief remained robust through the end of 2011, so for about 2.5+ years. He’s now first beginning to get some return of mild aching, but he can still exercise on a treadmill and walk extensively to play golf, things he couldn’t do before. As a result, at about the three year mark after his ankle stem cell treatment, we believe that because of the severity of his disease and his preexisting ankle fusion, it’s time for a stem cell treatment update. We’ll be seeing him in the Caymans for round two of his therapy. Given that he was an end stage patient when we began and that this ankle surgery alternative gave him his life back with only an injection, we feel he did very well. Having said that, we get asked by patients all the time, “How long will this last?” HK is an example where the results are life changing, but the full effects weren’t permanent.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.