SR: Low Back Disc Stem Cell Save
Like every procedure ever used in the history of medicine, ours has it’s successes and failures. For a physician, the successes are always more fun and some of those can get to be routine. However, every once in awhile you see a patient were the success was life changing. SR and her low back disc stem cell procedure is one of those wonderful successes.
I first blogged on her a few years back when I first saw her for SI joint problems. She was a real head scratcher, a young girl out of school due to back pain who was sidelined from competitive cheer-leading who had seen other physicians and tried lots of conservative care. Eventually I found a cyst on the front of her SI joint that was irritating the nerves going to her leg. We were able to treat the joint and get rid of the cyst and she promptly went back to school. This past year she returned to see me with severe back pain, again out of school and out of cheer-leading. I of course tried what had worked in the past, only to have it and everything else we tried like platelet epidurals, facet injections, and ligament injections fail. As a last resort, I noted that she had a disc bulge at L5-S1 on her more recent MRI that I felt could be causing the problem, but sending a young girl like this to surgery at 17 was sure to have her end up with a lifetime of back pain beginning in her 30s. This is when I suggested that her parents try the cultured disc stem cell technology we pioneered that’s now available in Grand Cayman.
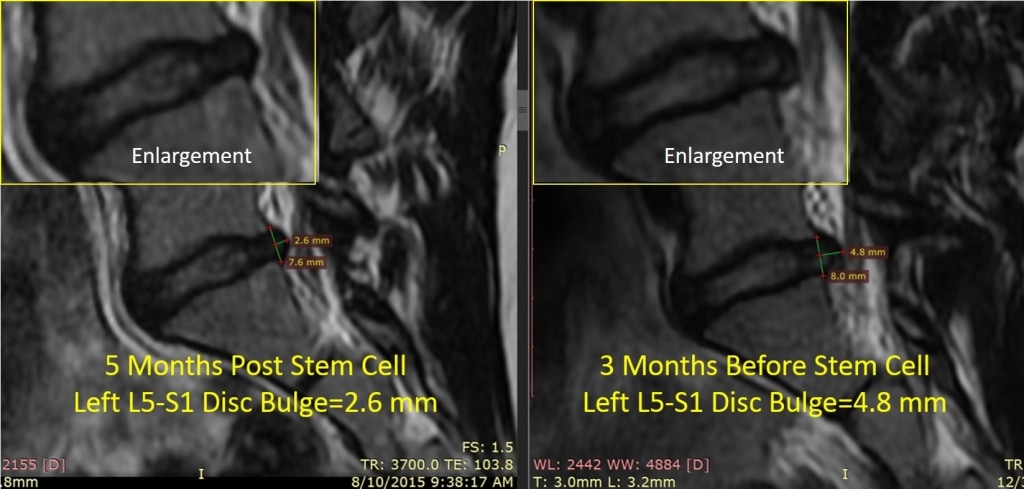
Above on the right is her pre-injection MRI showing a good sized disc bulge at L5-S1. On the left is her 5 month post stem cell injection image. The disc bulge that used to measure almost 5 mm is now about half it’s former size and her pain is down from an 8/10 to a 0/10. She’s back seeing a personal trainer to get back in shape and will start back in school in the fall.
The upshot? SR is one of those patients that I’m really happy we could help. She’s dodged the bullet twice now with regenerative medicine. We wish her a great school year!
The Regenexx-C procedure is not approved by the US FDA and is only offered in countries via license where culture expanded autologous cells are permitted via local regulations.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.