New Lab Research Doesn’t Help Improve Prolotherapy

Credit: Shutterstock
A colleague recently sent me a lab-based study that claims that the standard solution used for ligament and tendon prolotherapy should be lowered in concentration. Since that would be a big deal, I decided to take a deep dive into the study. What I found was that the study is not helpful in determining whether we should make that change.
What Is Prolotherapy?
Prolotherapy is the injection of a substance into a ligament or tendon which causes a brief inflammatory healing reaction. The goal is to give the damaged part another bite of the healing apple. There are a slew of things that can be injected that cause inflammation including hypertonic dextrose. This solution works because it’s more concentrated than the body’s tissues. This draws water out of the local cells and into the more concentrated solution, causing local cell injury and initiating an inflammatory response. This doesn’t last long as the body quickly deactivates this concentrated solution by water rushing into the place where it was injected. The local cell injury part is just a tiny fraction of the total cells, so minimal damage is done, just like surgically debriding a wound. Both chemical injection and surgical techniques signal the body that there is a new injury that needs to be repaired.
Note the following key parts of the dextrose prolotherapy process:
- A solution that is more concentrated (called hypertonic) than the body’s native concentration (which is 0.9%)
- Local cell injury caused by that solution
- The process of deactivation of the solution by water rushing toward the concentrated area to dilute the concentrated injection
- The local cell injury causes an inflammatory healing response
Lab Research on Prolotherapy is Challenging
Before I get into the new research that I feel is half-baked, note that because of the key components of how dextrose prolotherapy works, it would be almost impossible to study “in vitro” or in the lab. Why? because if you add hypertonic dextrose to cells in culture, the deactivation step will never happen. Why? That requires a fully living organism with a circulatory system, as the fluid needed to deactivate the prolotherapy solution isn’t available in the culture system.
At least two other studies have attempted to do this type of lab experiment in culture (i.e. not in a living organism). One also used hypertonic dextrose and human tendon human cells. Even without a circulatory system to bring in inflammatory cells, hypertonic dextrose did cause acute chemically mediated inflammation, which is what happens when cells are damaged (1).
The New Research
The new research we’ll review is from The Korean Journal of Pain and was published last year. It has a pretty impressive and certain sounding title: “The proper concentrations of dextrose and lidocaine in regenerative injection therapy: in vitro study” (2). Wow, as a physician reader, I expect that this will be the definitive study that tells me what concentrations of dextrose or lidocaine I should be using to treat my patients. Despite that title, as the British would say, the paper is “A few sandwiches short of a picnic”. Let’s dive in to see why.
First Problem-Wrong Cells
The researchers used a mouse embryonic fibroblast cell line. The first question is since the goal of the study is to define the proper concentrations of dextrose to be used in adult humans and not fetal mice, why use this cell line? We’ve performed studies with commercially bought human tendon cells, which are more expensive but widely available. So right off the bat, we have no idea what would happen if the study was repeated with human tendon or ligament cells.
Second and Fatal Problem-24 Hour Exposure to Hypertonic Dextrose
The researchers totally destroyed the ability of this study to apply to real-world prolotherapy by exposing the cells to hypertonic dextrose for a ridiculous period of time. Remember above that anytime a small amount of anything is injected into the body which is more or less concentrated than normal saline (0.9%) the laws of physics and fluid dynamics dictate that this injectate will be quickly brought into the normal concentration range by fluid moving in or out of the solution.
On a chemical basis, two items are at play here: diffusion and osmosis. Diffusion in water is codified by the second law of thermodynamics (entropy) which causes a more concentrated solution injected into a less concentrated solution to attract water molecules into itself to equilibrate the difference in concentration. The speed at which this happens is directly proportional to the concentration of the solution and the distance the water molecules need to travel. How fast does this happen? Just place red watercolor in a small vial of water and in minutes, the water in the red vial is uniformly red.
Osmosis is when there are semipermeable membranes involved, like cell walls. Here, these membranes only allow certain molecules like water to pass and also have specific pumps in them which can impact that phenomenon (i.e. they can pump sodium molecules one way or the other). However, the health of these walls is impacted by concentration. For example, once the water leaves the cell to dilute the injection of hypertonic solution, these pumps and the membrane are likely no longer functional as you now have a cell that looks more like a dehydrated raisin causing injury to the cell organelles.
This chemical phenomenon is also helped by the fact that any spot in the body can be overwhelmed with lots of fluid very quickly. For example, there is fluid in local cells getting contacted by the hypertonic injection. There are lymph channels in living tissues that also bring water in or out. Finally, hundreds of tiny arterioles (arteries) will also be in the vicinity also pumping fluid in. Hence, this process at most should take minutes.
In conclusion, local cells are never exposed to any hypertonic solution for anything close to 24 hours.
Our Research on the Impacts of Hypertonic Dextrose and MSCs
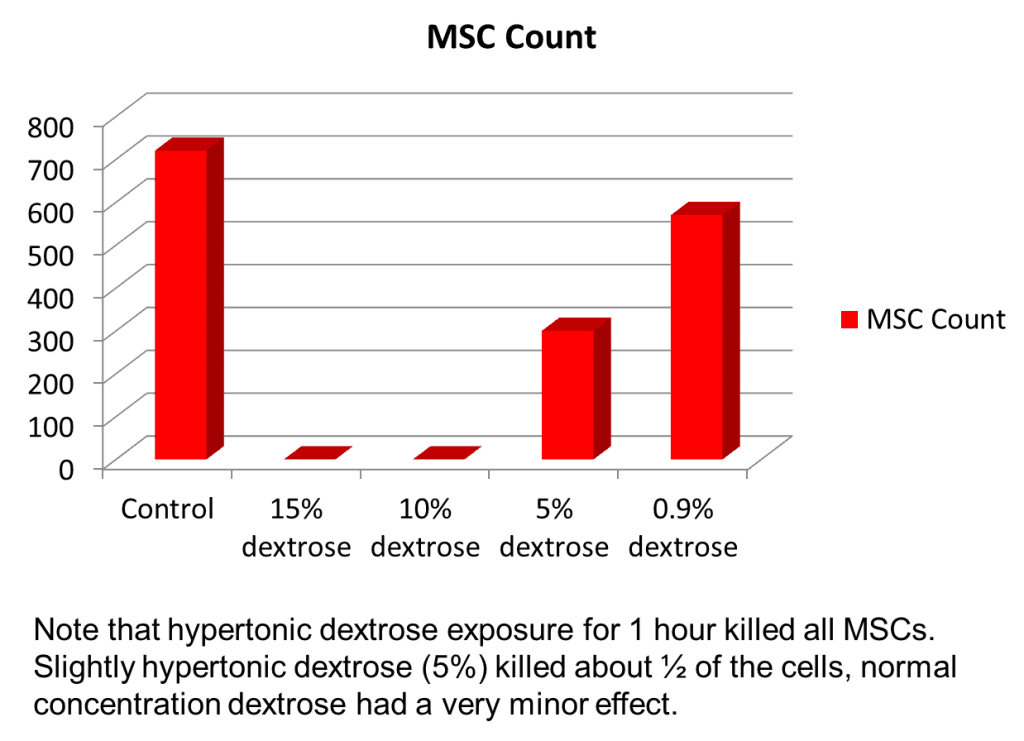
Our lab does lots of practical research that never gets formally published. This is on simple things like whether hypertonic dextrose impacts the growth factors in platelets (it doesn’t) or how using it at the same time as stem cells can injure those cells. We did the latter study many years ago because we saw a Chicago/Florida clinic combining prolotherapy and bone marrow concentrate. Rather than the crazy 24-hour exposure used in this study, we used only an hour for all of the reasons discussed in this blog.
That data is shown above. Note that the 5% dextrose killed about 60% of the stem cells and that 10 and 15% concentrations killed all of them. So while we learned that you can’t inject stem cells and dextrose prolotherapy solution together, since we were using a culture system like our Korean colleagues discussed here, we drew no conclusions on how this impacted prolotherapy in a living organism. For a clinical example, see how prolotherapy helped a patient avoid ACL surgery.
Animal Models
We do have research that shows that injecting 15% dextrose into the injured knee ligaments of rats improved the size of the ligament over saline injection and also made the ligament stronger (3). All other research that I found in animals used a different prolotherapy solution than dextrose. However, this rat study certainly suggests that what we see clinically, i.e. using 10-15% dextrose prolotherapy solution, works just fine in living organisms. Why?
In prolotherapy, you want a small amount of big cell injury because that causes the body to initiate an acute inflammatory healing reaction. Given that the hypertonic solution is then diluted and inactivated, the body then goes through its normal healing process over weeks and months in an environment unhindered by the initial hypertonic solution.
Lidocaine Toxicity
The authors did find out, like our research team, that concentrations of lidocaine above very minimal amounts are toxic to cells. We published on this concept many years ago, deciding that 0.25% of Ropivacaine is the max that should be used clinically and that clinicians should avoid using lidocaine and bupivacaine (4). However, even those conclusions could be impacted by the concentration diluting effects I discussed above. Meaning, if you inject 0.5% Ropivacaine into a living organism with a circulatory system, how long doe that concentration remain at 0.5%?
Should We Go Lower on Dextrose Concentrations?
We’ve observed and published real-world objective evidence in traumatic neck injury patients that using 12.5% dextrose works to tighten ligaments (5). Hence, there isn’t a big practical impetus to want to change what works. However, as shown here, any study looking at using less concentrated hypertonic dextrose can’t reliably be done in the lab but would require a real animal with a circulatory and immune system. To perform that study, you would need an animal model injected with various dextrose concentrations. As far as I know, no study like that exists.
The upshot? Regrettably, this new research on prolotherapy concentrations doesn’t add much to figuring out the right concentration of dextrose to use for this procedure in real patients. Hence, I await more research on this topic.
___________________________________________________
References:
(1) Ekwueme EC, Mohiuddin M, Yarborough JA, Brolinson PG, Docheva D, Fernandes HAM, Freeman JW. Prolotherapy Induces an Inflammatory Response in Human Tenocytes In Vitro. Clin Orthop Relat Res. 2017 Aug;475(8):2117-2127. doi: 10.1007/s11999-017-5370-1. Epub 2017 Apr 27. PMID: 28451864; PMCID: PMC5498388.
(2) Woo MS, Park J, Ok SH, Park M, Sohn JT, Cho MS, Shin IW, Kim YA. The proper concentrations of dextrose and lidocaine in regenerative injection therapy: in vitro study. Korean J Pain. 2021 Jan 1;34(1):19-26. doi: 10.3344/kjp.2021.34.1.19. PMID: 33380564; PMCID: PMC7783851.
(3) Jensen KT, Rabago DP, Best TM, Patterson JJ, Vanderby R Jr. Response of knee ligaments to prolotherapy in a rat injury model. Am J Sports Med. 2008;36(7):1347-1357. doi:10.1177/0363546508314431
(4) Dregalla RC, Lyons NF, Reischling PD, Centeno CJ. Amide-type local anesthetics and human mesenchymal stem cells: clinical implications for stem cell therapy. Stem Cells Transl Med. 2014 Mar;3(3):365-74. doi: 10.5966/sctm.2013-0058. Epub 2014 Jan 16. PMID: 24436443; PMCID: PMC3952925.
(5) Dregalla RC, Lyons NF, Reischling PD, Centeno CJ. Amide-type local anesthetics and human mesenchymal stem cells: clinical implications for stem cell therapy. Stem Cells Transl Med. 2014 Mar;3(3):365-74. doi: 10.5966/sctm.2013-0058. Epub 2014 Jan 16. PMID: 24436443; PMCID: PMC3952925.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
