Regenative Labs Sues CMS and the Entire Medicare Program

Credit: Shutterstock
As I have reviewed in this blog, Medicare recently issued a massive clawback likely worth about half a billion dollars for payments made for birth tissue products to treat problems like knee arthritis and pain. This week, one of the companies involved called Regenative Labs has sued CMS and all of its national Medicare contractors in DC federal court. Let’s dig in.
Before We Get Into It
I am not an attorney, but a doctor. While I have followed these issues closely for years and have direct experience with how the FDA regulates this space and how they react to various actions, this blog represents my personal opinions.
What’s Going On?
Through the years, this blog has covered the myriad problems with amniotic and umbilical cord tissue vendors. At first, these companies claimed they were selling products that contained millions of live and highly functional mesenchymal stem cells. Based on the published peer-reviewed literature (including our study) this was FICTION. Despite the lack of functional stem cells in these products, these claims were responsible for what in my opinion was massive consumer fraud committed by countless clinics who sold these as stem cell products to patients.
Next, IMHO, as if pretending that their products were stem cell laden wasn’t bad enough, many of these companies jumped on the Medicare Q-code bandwagon. What’s that?
These companies applied for reimbursement codes (called Q-codes). Some of the companies were granted these codes in error (according to Peter Marks of the FDA) but there were never any Medicare guidelines that stated that Medicare should reimburse these codes. How does that work?
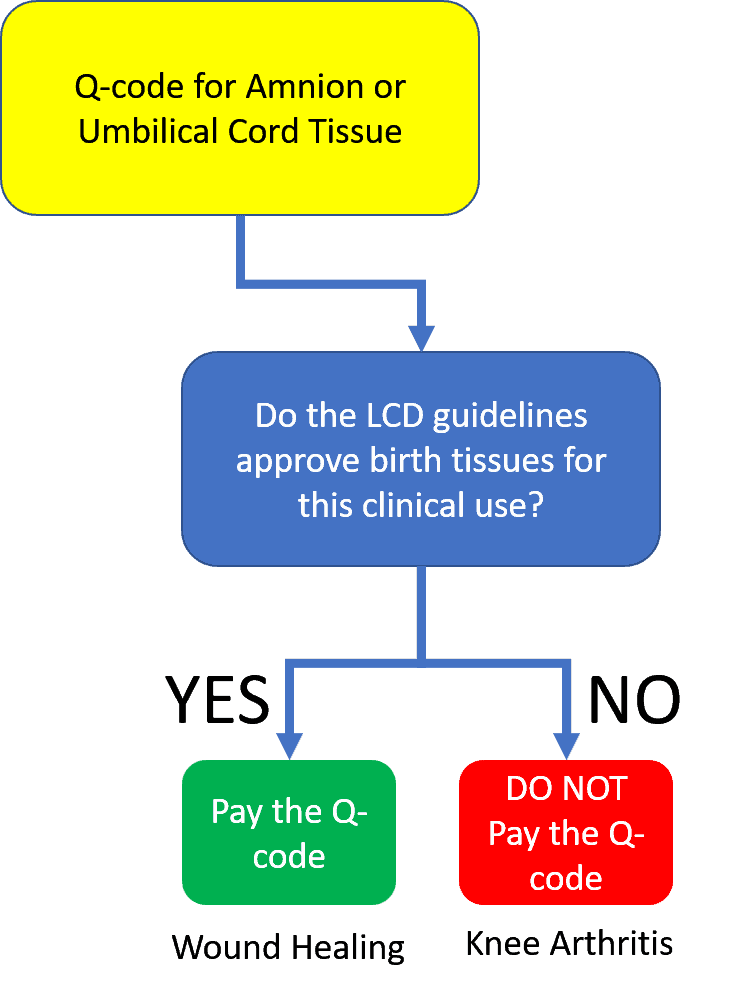
As you can see above, getting paid for the use of a product like this requires a two-step process. One is a Q-code and the other is a local Medicare guideline (called a “Local Coverage Determination” or LCD) that states that Medicare covers your birth tissue product for this specific medical problem (like knee arthritis). These companies had the first step done, but the LCDs stated that there was no coverage.
Despite that, the companies hired billing companies to bypass or appeal Medicare denials, which resulted in hundreds of millions being paid to providers. Once Medicare got wind of all of this, they issued one of the biggest clawbacks in the program’s history. Meaning Medicare now wants all of that money back. In addition, these huge clawbacks usually come with somebody going down with criminal charges, so that shoe has yet to drop.
Who is Regenative Labs?
Regenative Labs is a company that sells umbilical cord tissue products called ProText and CellText. I have blogged on this company a few times:
- In 2020, Regenative claimed to be selling a mesenchymal stem cell product that could treat things like arthritis and pain
- In 2021, I was sent a sales rep’s email that claimed that Regenative’s products could be reimbursed by Medicare after it became clear that the justice department was collecting data through Civil Investigative Demands on a possible clawback
The Lawsuit
On 3/15/22 Regenative sued the entire Medicare system in the Washington DC district court. Do you think I’m kidding? Here’s the list of entities sued:
- HHS
- CMS
- Noridian (Medicare contractor)
- WPS (Medicare contractor)
- NGS (Medicare contractor)
- CGS (Medicare contractor)
- Palmette0 (Medicare contractor)
- First Coast (Medicare contractor)
Regenative’s Position
The basic Regenative argument is that Medicare has determined that Regenative’s products should be regulated as a 351 drug (when used to treat pain conditions) and thus are not eligible for reimbursement because they require FDA approval before clinical use. Regenative asserts that this determination can’t be made by CMS and could only be made by the FDA. Also, if Medicare had wanted to extend its powers in this way, the agency would have to either change the law or go through “comment and rulemaking” to allow its own rules to determine what was and was not a cell-based drug. Regenative also states that it will basically go out of business if the judge doesn’t act now to stop Medicare from suspending payments and clawing back all of the money it paid for the company’s products.
361 vs 351?
One of the things you need to understand to comprehend this argument is the HUGE difference between a 361 tissue and a 351 cell based drug. The FDA has a simple “honor system” style registration for companies that sell tissue transplant products also called a “361 product”. This doesn’t require any FDA approval or review, so you can literally get your product to market in 45-minutes online.
One of the problems faced by birth tissue vendors is that the FDA system classifies products based on the claims made. So the moment you begin to claim that you’re selling a stem cell product instead of just lifeless umbilical cord tissue, all bets are off and you can no longer use the 361 system and must go through the 351 “cell drug” pathway. That requires extensive clinical trials, often hundreds of millions of dollars, and many years to clear all of those hurdles. Hence, no birth tissue product to date has been approved by FDA to treat pain or arthritis.
As you might imagine, the HUGELY lowered bar for a 361 tissue registration has the effect of companies self-classifying themselves this way because it’s far faster and cheaper. Hence, the FDA has had to send out numerous Warning and Untitled Letters to companies who claimed to be selling or using 361 umbilical cord tissue that the agency later determined to be an unapproved 351 drug:
Hence, IMHO, anybody paying attention could see from these FDA letters that claiming to sell an umbilical “stem cell” product as a 361 was a fool’s errand. In addition, FDA made it even clearer in a 2019 consumer warning:
Is Regenative Selling a 361 Tissue or an Unapproved 351 Drug?
This entire case, in my opinion, will turn on two critical points:
- 361 or 351?
- FDA’s involvement in issuing Q-codes
361 or 351?
First, Regenative claims that it’s selling a 361 tissue, hence CMS can’t stop or clawback payment because the agency has determined that it’s actually selling an unapproved 351 drug product. From the lawsuit:
- “Since beginning operations, Regenative has taken all steps necessary to ensure that
its products, Coretext and Protext, satisfy the elements required to be marketed, distributed, and regulated solely under Section 361, including registering with the FDA, listing its HCT/Ps with the FDA, following legal and scientific guidance, and applying for and receiving a Q-code from CMS (Q4246), which expressly recognized Regenative’s products are 361 products.”
Is this a true assertion that Regenative is selling a 361 tissue? Remember, all it takes to be classified as a drug is a claim that you’re selling a stem cell product. I took these pictures of Regenative’s booth at a recent medical conference:
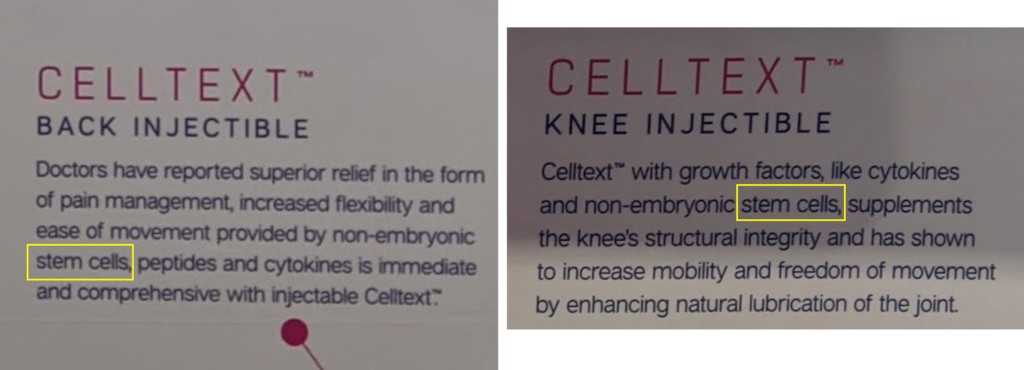
The above pictures show that Regenative was selling a stem cell product to treat back and knee pain. That claim alone, regardless of how Regenative processes its tissues, IMHO makes these products unapproved 351 drugs. Hence, Regenative fails on that point.
FDA’s involvement in issuing Q-codes
The other major part of this lawsuit is the idea that CMS acted alone when it classified Regenative’s products as a 351 drug. The company states that CMS doesn’t have that authority. However, the problem with that assertion is that the FDA has already publically discussed its failure in working with CMS to block Q-codes being granted in error.
This is what Regenative Labs states in its lawsuit:
- “Defendants, via the Policy, invade the statutory authority of the FDA by requiring
all placental tissue products, including Regenative’s Section 361 products, to become classified as and receive Pre-Market FDA Approval under Section 351. - “To date, the FDA has never rejected or otherwise indicated any disagreement with the designation of Regenative’s products as being properly regulated solely under Section 361.”
- “The determination as to whether a product is regulated as an HCT/P under Section 361 or a biologic or drug under Section 351 is made by the FDA, not CMS.”
The problem here, as someone who has watched this issue closely, is that the FDA has already stated that it was asleep at the switch when it comes to this Q-code problem. Meaning the agency has a first right of refusal to block companies applying for CMS Q-codes that haven’t gotten the FDA’s blessing. In this way, the two agencies work hand in glove.
How do we know this? Because Peter Marks said it in an online medical conference.

Peter Marks is the head of the FDA division (CBER) that regulates Regenative Labs and its products. Here is a transcript of what he said at a May 2021 AAOM medical conference via Zoom.
Here is the question as read by Dr. Marks:
“Companies claiming that their amniotic products have an approved Q-code to be reimbursable to treat orthopedic conditions.”
Peter Marks Answer: “We are currently in the process of rectifying that. We’ve met with CMS and it turns out that it’s not trivial to get rectified but it is an issue and these should not have a Q-code for orthopedic conditions, because it’s not consistent, it can only be used consistent with the way we would use amniotic tissues which is as a covering.”
Later after someone else brings this same topic up…
“This Q-code issue is something that I apologize for….let’s be transparent. This is what happens in big bureaucracies in government. You have CMS over here and FDA over here, the companies apply to CMS, CMS doesn’t talk To FDA and so some of this Q-code stuff has occurred. We’re currently, I can’t tell you how and when it’s going to be finished cleaning up, but we are currently in the middle of a clean-up process there and it may take a little time to get there but the correct answer…”
Hence, the FDA chief that regulates Regenative Labs clearly says that they should have been involved with CMS when these codes were issued and that they apologize for screwing that up. He also says they are trying to fix it.
What Will Likely Happen with this Lawsuit?
Based on my direct experience and in my opinion, this lawsuit will be thrown out at the motion to dismiss phase. Why? Because the courts give CMS and other government agencies a wide birth to interpret their own rules and regulations as they see fit. Getting over that HUGE barrier to claim that CMS violated its own rules is very hard. Only a tiny fraction of such lawsuits against government agencies are able to get past this early step.
If Regenative gets past the government’s motion to dismiss, it has another HUGE hurdle. The claim that Regenative is selling a 361 tissue is hard to digest when it had a booth at a medical conference that claimed to be selling stem cell products to treat knee arthritis and pain. In addition, it’s hard to believe that the FDA will back Regenative when the agency’s existing communications on umbilical products all point in the direction of these products being 351 drugs. Frankly, all FDA needs to do is to publish a Warning or Untitled Letter that ProText and CellText are 351 drugs and it’s game over.
The Bigger Risk
In my opinion, there’s an even bigger risk here. It’s one thing to disagree that your product should be regulated as a tissue or a drug, but it’s yet another to do that in the context of Medicare billing. Remember, submitting knowingly false or inaccurate claims to Medicare to get the system to spit out payments has very real criminal teeth. That’s ten years in federal prison for each fraudulent bill submitted. Hence if CMS wants to play rough here, they only need to convince the justice department that the companies that used Q-codes did so with full knowledge that they were gaming the system and criminal charges will follow. So in my opinion, this is one VERY high-risk game of chicken.
The upshot? In my opinion, this lawsuit will fail on several different points. However, I certainly admire Regenative’s Moxy even if I don’t admire the company. In the end, it will be interesting to see what happens as this massive clawback winds its way through the system. Will that result in Regenative going out of business as it claims? Given that they have asked for the judge to quickly step in and issue an injunction against the clawback, we should have some sense of where all of this is headed very soon.

NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.
